(a) Complete the series of reactions in Fig. P19.50 by giving the major organic product. (b) Show...
Question:
(a) Complete the series of reactions in Fig. P19.50 by giving the major organic product.
(b) Show how the same product could be prepared from hydroquinone monomethyl ether (p-methoxyphenol).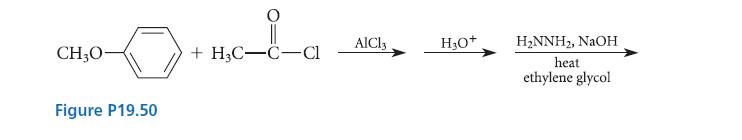
Transcribed Image Text:
CH₂0- Figure P19.50 +H3C-C-Cl AlCl3 H₂O+ H₂NNH₂, NaOH heat ethylene glycol
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
a FriedelCrafts acylation gives a ketone pmethoxyacetophenone which reacts i...View the full answer

Answered By

Kennedy Odhiambo
As a professional writer, I have been in the field for over 5 years having worked as a lecture in different tertiary institutions across the world. With this impeccable experience, I assure provision of a good and supporting environment for students to learn.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Show how the following compounds could be prepared from the given starting materials. You can use any necessary organic or inorganic reagents. a. b. c. d. e. f. CH3CH2CNH2 C CH.CH-CH-CH-OH-....
-
Show how the following compounds could be prepared from benzene: a. b. c. CH2 OCH3 Br NO2 CH3 SO H CH2CH CH3)2
-
Show how the following compounds could be prepared from cyclohexanone 0 CCH2CH2CH3 CH CCH3 CH2CH2CH3 CH CH2CH3 CH3
-
The profit before tax as reflected in the draft statement of comprehensive income of Sword Limited for the financial years ended 31 December 2020 and 31 December 2021 respectively was as follows:...
-
After closing its accounts at July 31, 2012, Goodrow Electric Company had the following account balances: Requirement 1. Prepare Goodrows post-closing trial balance at July 31,2012. Long-term...
-
An analyst estimates that the probability of default on a seven-year AA-rated bond is 0.06, while that on a seven-year A-rated bond is 0.13. The probability that they will both default is 0.04. a....
-
Two conspirators, Timothy Carpenter and Timothy Sanders, were charged with aiding and abetting a robbery affecting interstate commerce and the use or carriage of a firearm. At trial, the FBI...
-
Three different plans for financing a $200,000,000 corporation are under consideration by its organizers. Under each of the following plans, the securities will be issued at their par or face amount,...
-
Instructions: Place the six steps of the accounting cycle in the correct order. Rank the options below. Prepare financial statements.Prepare financial statements. open choices for ranking No answer...
-
Which of the following is not a good thing to do during an interview? a. Conduct the interview in private. b. Establish the purpose of the interview. c. Interview more than one person at once. d. Do...
-
Using known reactions and mechanisms discussed in the text, complete the reactions given in Fig. P19.49. (a) (b) i (CH3CHCH)C-C-CHCH3 PhP i CH3CHCHCCH3 + HN-Ph NaBH4 CH3OH NaH NaBH4 CH3OH
-
Find the Maclaurin series of f (by any method) and its radius of convergence. Graph f and its first few Taylor polynomials on the same screen. What do you notice about the relationship between these...
-
After graduation from METU you will start working as an operations manager at an electronics manufacturer that faces assembly-line balancing problems. You have been told that the firm needs to...
-
An organization's portfolio investment process consists of the following activities performed once a year: ideas for projects are collected along with their projected benefits, development managers...
-
You just acquired a mortgage in the amount of $249,500 at 5.75 percent interest, compounded monthly. Equal payments are to be made at the end of each month for thirty years. How much of the first...
-
An atom has a diameter of 3.00 A and the nucleus of that atom has a diameter of 7.50 10 5 . Determine the fraction of the volume of the atom that is taken up by the nucleus. Assume the atom and the...
-
1. Why are people in India prosperous everywhere else besides India? What is the evidence? 2. Why are people from Hong Kong so prosperous as compared to everywhere else? What is the evidence? 3. What...
-
Knickknack, Inc. manufactures two products: odds and ends. The firm uses a single, plant wide overhead rate based on direct-labor hours. Production and product-costing data are as follows:...
-
Design a circuit which negative the content of any register and store it in the same register.
-
Dianeackerone is a volatile natural product isolated from secretory glands of the adult African dwarf crocodile. The compound is believed to be a pheromone associated with nesting and mating....
-
Outlined here is a synthesis of glyceraldehyde (Section 5.15A). What are the intermediates A-C and what stereoisomeric form of glyceraldehyde would you expect to obtain? PCC CH3OH, HA KMnO4, HO H2o ...
-
Consider the reduction of (R)-3-phenyl-2-pentanone by sodium borohydride. After the reduction is complete, the mixture is separated by chromatography into two fractions. These fractions contain...
-
Example Problem 2.2 The self-ignition temperature (SIT) of a fuel is, as discussed in Chapter 6, the temperature at which the fuel will self-ignite. A diesel engine will not start unless the...
-
An uncoated steel (assumed to be iron for simplicity) pipeline is proposed for the transfer of a deaerated weak acidic solution. Define the anodic and cathodic reactions. Calculate the equilibrium...
-
12.) A vertically mounted double acting hydraulic cylinder (with the rod end down) is used to raise and lower a heavy load, as well as safely stop and hold it (load lock) in any position. Complete a...

Study smarter with the SolutionInn App


