A student has run the reactions shown in Fig. P11.51 and is disappointed to find that each
Question:
A student has run the reactions shown in Fig. P11.51 and is disappointed to find that each has given none of the desired product. Explain why each reaction failed.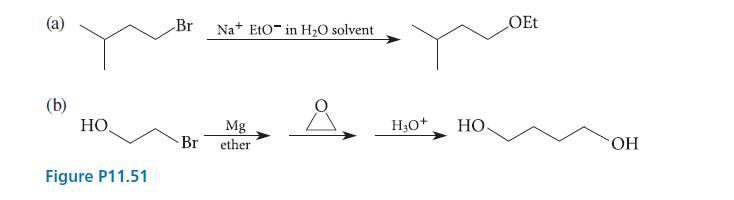
Transcribed Image Text:
(b) HO, Figure P11.51 Br Na EtO in H₂O solvent Mg Br ether H3O+ HO OEt OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b Sodium ethoxide reacts with water to give ethanol and sodium hydroxide Although the pK ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A student who has created a linear model is disappointed to find that her R2 value is a very low 13%. a) Does this mean that a linear model is not appropriate? Explain. b) Does this model allow the...
-
Leadership in Organizations Module Three Assignment Essay #1: Case Study: Echo Electronics Paul Sanchez is the production manager for Echo Electronics, a small company that makes and distributes...
-
An intern who has created a linear model is disappointed to find that her R2 value is a very low 13%. a) Does this mean that a linear model is not appropriate? Explain. b) Does this model allow the...
-
The preparation of an organization's budget: a. forces management to look ahead and try to see the future of the organization. b. requires that the entire management team work together to make and...
-
Suggest reasons why the total assets and total liabilities of a defined benefit pension plan do not appear, but their net amount does appear, on the employers balance sheet.
-
Use the statements of earnings for Campbell Soup Company to perform a vertical analysis for each item reported for the last two fiscal years using net sales as the base. What does your analysis tell...
-
If your instructor assigns a marketing plan for your class, we hope you will be excitedfor two reasons. First, you will get insights into trying to actually do marketing that often go beyond what you...
-
Booker Co. sold $300,000, 10%, 10-year bonds on January 1, 2014. The bonds were dated January 1, and interest is paid on January 1 and July 1. The bonds were sold at 104. Instructions (a) Prepare the...
-
A small electric immersion heater is used to heat 87 g of water for a cup of instant coffee. The heater is labeled "120 watts" (it converts electrical energy to thermal energy at this rate)....
-
When HCl is formed as a by-product in reactions, it is usually removed from reaction mixtures by neutralization with aqueous base. At times, however, the use of base is not compatible with the...
-
Give the major organic product of each of the following reactions. Include stereochemistry where relevant. (a) Dibutyl sulfide with 1 equivalent of H 2 O 2 at 25C (b) Dibutyl sulfide with 2 or more...
-
What are a firms major motives in the decision to establish manufacturing facilities in a foreign country?
-
If X is a normal variable with known variance equal to 650, how large a sample must we take to be 90 % confident that the sample mean will not differ from the true mean by more than 5 units?
-
Suppose you are working for a political consulting firm that is trying to forecast the outcome of a presidential election. Because of the time and cost involved in conducting a simple random sample...
-
Suppose a quality control expert is interested in drawing a random sample of 100 light bulbs from a case of 10,000. Explain how a table of random numbers could be used to do this.
-
The academic advisor to the football team of Rah Rah University is interested in the mean number of hours that players spend studying during the football season. Of the 100 members of the football...
-
The Mom and Pop Grocery Store has 115 employees. In a random sample of 30 of these employees, the mean number of days that an employee was late each year was 14 days, and the sample standard...
-
Are all losses realized on the sale of capital assets deductible?
-
Apply Jacobis method to the given system. Take the zero vector as the initial approximation and work with four-significant-digit accuracy until two successive iterates agree within 0.001 in each...
-
Which amide bonds in the following polypeptide are cleaved by trypsin by chymotrypsin? Phe-Leu-Met-Lys-Tyr-Asp-Gly-Gly-Arg-Val-IIe- Pro-Tyr
-
What kinds of reactions do the following classes of enzymes catalyze? (a) Hydrolases (b) Lyases (c) Transferases
-
Which of the following amino acids are more likely to be found on the outside of a globular protein, and which on the inside? Explain. (a) Valine (b) Aspartic acid (c) Phenylalanine (d) Lysine
-
A house flipper found a diamond in the rough and, after buying it at a sheriff's auction, has identified six systems that need significant work. The estimated completion times (in weeks) and costs...
-
Work Opportunity Tax Credit (WOTC). (Obj. 3) In August of 2021, Arthur Co. hired Jan, who is a long-term family assistance program recipient. Jan's qualified first-year and second-year wages were...
-
Finished goods inventory on September 30, 2019 was 3,000 units. Projected sales for January are 25,000 units. The sales price is $100 per unit. The company tries to keep ending finished goods...

Study smarter with the SolutionInn App


