(a) Write the equations for the first and second ionizations of succinic acid. Label each with the...
Question:
(a) Write the equations for the first and second ionizations of succinic acid. Label each with the appropriate pKa values from Table 20.3.
(b) Why is the first pKa value of succinic acid lower than the second pKa value?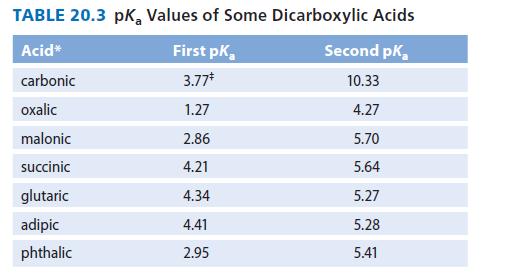
Transcribed Image Text:
TABLE 20.3 pk, Values of Some Dicarboxylic Acids Acid* Second pk carbonic 10.33 oxalic 4.27 malonic 5.70 succinic 5.64 glutaric 5.27 adipic 5.28 phthalic 5.41 First pK, 3.77* 1.27 2.86 4.21 4.34 4.41 2.95
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a The first and second ionizations of succinic acid H0 HOCCHCHCOH HO PK 421 OCCHCHCOH HO HO PK564 ...View the full answer

Answered By

Shantanu Salokhe
Hi! My name is Shantanu. I have 2 years experience in the teaching field. My focus is to help communicate & breakdown difficult concepts in the way that allows students greater accessibilities & comprehension to their course material. I Excel in the categories of maths & civil engineering. I assure you my knowledge will be helpful for you. I will help you as much as I can!
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Malonic acid has two carboxylic acid groups and consequently undergoes two ionization reactions. The pK a for the first ionization of malonic acid is 2.86; the pK a for the second is 5.70. The pK a...
-
A liquid mixture contains N components (N may be any number from 2 to 10) at pressure P(mm Hg). The mole fraction of the ith component is x i (i = 1. 2...., N), and the vapor pressure of that...
-
Write the equations for the removal of one amino acid from the peptide alanylglycylvaline by the Edman method. What is the name of the remaining dipeptide?
-
Suggest two reasons why the adjustments proposed by independent auditors more often than not call for reducing recorded earnings.
-
Selected accounts of Blume Irrigation System at December 31, 2012, follow: Requirements 1. Prepare the companys classified balance sheet in report form at December 31, 2012. 2. Compute the companys...
-
The monthly closing stock prices (rounded to the nearest dollar) for Starbucks Corp. and Panera Bread Co. for the first six months of 2010 are reported in the following table. a. Calculate the sample...
-
I spent time with you. It seems that this deed is out of character for you. You were not awarded your normal annual bonus. I would probably feel the same. Is that what happened? You normally wouldnt...
-
Messner Co. reported $145,000 of net income for 2010. The accountant, in preparing the statement of cash flows, noted several items occurring during 2010 that might affect cash flows from operating...
-
Jacinta had a bad experience with an insurance agent when her house flooded. The insurance agent was generally unhelpful and rude. She found herself becoming distrustful of all insurance agents,...
-
Give the structure of the compound with molecular mass = 88 and the following spectra. Proton NMR: 1.2 (6H, , J = 7 Hz); 2.5 (1H, septet, J = 7 Hz); 10 (1H, broad s) IR: 26003400 cm 1 (broad),...
-
At a given concentration of acetic acid, in which solvent would you expect the amount of acetic acid dimer to be greater: CCl 4 or water? Explain.
-
Cantelevellers plcs primary financial objective is to maximise the wealth of its shareholders. The business specialises in the development and assembly of high quality television sets. It normally...
-
Use the table to calculate the GDP Deflator for 2021. Use 2018 as the base year.
-
A mass m = 1,4 kg ball is swinging in a vertical circle of radius r = 22 cm. If the constant speed of the ball is v=6,6 m/s. Determine the tension in the string at the top of the circle in units of N...
-
The Botosan Factory has determined that its budgeted factory overhead budget for the year is $586,245 and budgeted direct labor hours are 313,500. If the actual direct labor hours for the period are...
-
Adam Holmes is the Processing Manager of Empire Mortgage Company, a firm that processes loan applications for a number of regional builders. Home buying and therefore mortgage processing is a highly...
-
A gas expands by exerting a constant pressure of 2 atm from a volume V 1 = 2 0 0 cm 3 to V2 = 5 0 0 cm 3 . What work does the gas do ?
-
Use the Internet to access the Web site for Weyerhaeuser (www.we.yerhaeuser.com). International Paper (www.intenationalpaper.com) or Boise Cascade (www.boisccascade.com). Required: Skim over the...
-
Sandcastles, Inc.s management has recently been looking at a proposal to purchase a new brick molding machine. With the new machine, the company would not have to buy bricks. The estimated useful...
-
Starting with aniline, outline a synthesis of each of the following: (a) p-Bromoaniline (b) o-Bromoaniline (c) 2-Bromo-4-nitroaniline (d) 4-Bromo-2-nitroaniline (e) 2,4,6-Tribromoaniline
-
Both of the following syntheses will fail. Explain what is wrong with each one. (a) (b) NO2 (1) HNO/H SO (2) CH3COC/AIC (1) NBS, light (2) NaOEt, EtOH, heat (3) Br2, FeBr3
-
Propose structures for compounds G-I: concd HNO heat 60-65 OH
-
1. Prove that: (25p) [A (Bx C)] + [B( A)] + [C( B)] = 0 2. Three vectors A, B and C are given by: A = 3x-2+22 B=6x+4y-22 C=-3x-2-42 Compute the values of A. Bx C and A (B C) and C(A B) and Bx( A)....
-
Find the area of each. 1) 60 ft 15 yd 10.5 yd
-
You have sampled the soil of your garden and it has a soil organic carbon (SOC) content of 5.39 % SOC. Our class notes on organic matter give a simple method for converting between SOC and soil...

Study smarter with the SolutionInn App


