Alkenes undergo the addition of thiols at high temperature in the presence of peroxides or other free-radical
Question:
Alkenes undergo the addition of thiols at high temperature in the presence of peroxides or other free-radical initiators. The following reaction is an example.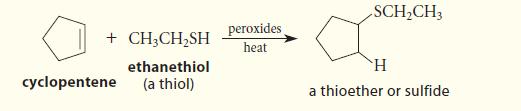
Propose a mechanism for this reaction.
Transcribed Image Text:
+CH3CH₂SH ethanethiol (a thiol) cyclopentene peroxides heat SCH₂CH3 H a thioether or sulfide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The fact that the reaction requires peroxides tells us that a freeradical mechanism is operating The initiation step is abstraction of a hydrogen atom ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Thiols can be prepared from the reaction of thiourea with an alkyl halide, followed by hydroxide-ion-promoted hydrolysis. a. Propose a mechanism for the reaction. b. What thiol would be formed if the...
-
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of a peroxide. ROOR- H2C CHCH high pressure C-C propylene I7 polypropylene
-
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of benzoyl peroxide. H CH n HC-CH CH3 propylene high pressure polypropylene
-
Suppose that Serendipity Bank has excess reserves of $8000 and checkable deposits of $150,000. If the reserve ratio is 20 percent, what is the size of the banks actual reserves?
-
Fresh Foods, a large restaurant chain, needed to determine if it would be cheaper to produce 5,000 units of its main food ingredient for use in its restaurants or to purchase them from an outside...
-
Comment on the following statement: The conditions for foreign buyers interested in U.S. targets could not be more auspicious. The dollar is weak, M&A financing is harder to come by for financial...
-
Assuming the same data as given in problem 9, was the well in each case profitable? Discuss your answer. Problem 9:- Property cost (acquisition cost). Drilling cost (one well). Estimated completion...
-
Beacon Company maintains and repairs warnings lights, such as those found on radio towers and lighthouses. Beacon Company prepared the end-of-period spreadsheet shown on the next page at October 31,...
-
Find the two unknown sides in this triangle. Keep 4 significant digits. x+3.10m -240m-x 5.21m
-
Estimate the standard enthalpies of the reactions shown in Eqs. 5.67 and 5.68. (CH3)3C-O + H-Br tert-butoxy radical (CH3)3C-O-Br + .H (5.68)
-
Give the structure of the alkene that could be used as a starting material to form chlorohydrin B in Study Problem 5.1. STUDY PROBLEM 5.1 Which of the following chlorohydrins could be formed by...
-
Find the indefinite integral. csc 2 x /cot 3 x dx
-
ABC Co has a target debt ratio of 40% and it keeps this target. The cost of equity is 10% and the cost of debt is 4%. It is considering expanding its business and requires $1 million investment....
-
Consider an economy where there is uncertainty about the future. In particular, at the current date t all agents are unsure which of the 3 possible states (w1, W2, W3) will mate- rialize at date t+...
-
A number of investment strategies based on risk factor research have been lumped under the heading "smart beta," but are they smart? What are the arguments for and against smart beta strategies and...
-
The pharmacy's computer system costs $ 950,000 aA pharmacy purchased a bottle of Amoxil for $27.14 and sold it for $42.80. What was the % profitend should last 5 years. Its disposal value is $55,000....
-
Briefly describe the three main processes that utilize ATP as a free energy source.
-
Portions of the financial statements for Hawkeye Company are provided below. Required: 1. Prepare the cash flows from operating activities section of the statement of cash flows for Hawkeye Company...
-
Suppose the S&P 500 futures price is 1000, = 30%, r = 5%, = 5%, T = 1, and n = 3. a. What are the prices of European calls and puts for K = $1000? Why do you find the prices to be equal? b. What...
-
Draw the structures of these compounds: (a) 4-Methylocatane (b) 2, 4-Dimethyl-5 propyl decane
-
What is wrong with these names? Provide the correct name for each. (a) 5, 5 Dimethyl-3-ethylhexane (b) 2-Dimethyl pentane
-
Provide name for these complex groups: CHCHCH3 a) -CHCHCH3 CH3 b) CHCHCH CH, c) -CHCCH3 CH3
-
For small amplitude oscillations (deviation from vertical of less than about 10), the motion of a simple pendulum is very close to that of a simple harmonic oscillator. The expression for the period...
-
X-ray tube. An x-ray tube is an evacuated glass tube that produces electrons at one end and then accelerates them to very high speeds by the time they reach the other end. The acceleration is...
-
2. A screen is separated from a double-slit setup by a distance of 2.15 m. The slit spacing is 0.0150 mm. The first-order maximum is 4.05 cm from the centerline. Find (a) the wavelength, (b) the...

Study smarter with the SolutionInn App


