Although enols are unstable compounds (Sec. 14.5A), suppose that the acidity of an enol could be measured.
Question:
Although enols are unstable compounds (Sec. 14.5A), suppose that the acidity of an enol could be measured. Which would be more acidic: enol A or alcohol B? Why?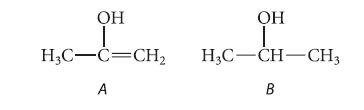
Transcribed Image Text:
OH OH I H3C-C=CH₂ H3C-CH-CH3 A B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The enol A is more acidic by about five pK a units ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
There are four diastereomers (A D, margin) of (4S)-2-bromo-4-phenylcyclohexanol. As a team, formulate their structures and draw each diastereomer in the most stable chair conformation (see Table...
-
The following Excel output summarizes the results of an analysis of variance experiment in which the treatments were three different hybrid cars and the variable measured was the miles per gallon...
-
Use the chart of accounts you created in Chapter 1 (and add accounts where necessary) All of the first months activity for Shine King Cleaning is as follows. Nov 1 Evan Hudson deposited $35,000 in...
-
In the planning stage, a sample proportion is estimated as = 40/50 = 0.80. Use this information to compute the minimum sample size n required to estimate p with 99% confidence if the desired margin...
-
Five projects form the mutually exclusive, collectively exhaustive set under consideration. The cash flow profiles for the five projects are given in the table below. Information on each project was...
-
Emily Jackson (Social Security number 765-12-4326) and James Stewart (Social Security number 466-74-9932) are partners in a partnership that owns and operates a barber shop. The partnerships first...
-
Two blocks are fastened to the ceiling of an elevator as in Figure. The elevator accelerates upward at 2.00 m/s. Find the tension in each rope. T 10.0 kg T 10.0 kg
-
Enols, like phenols, have pK a values of ~1011. However, the pK a of warfarin, a widely used anticoagulant, is 5.0. (a) Give the structure of the conjugate base of warfarin. (b) Use resonance...
-
Give the products formed in the reaction of 1-hexene with each of the following compounds in the presence of the Grubbs G2 catalyst. (a) An excess of allyl alcohol (2-propen-1-ol) (b) An excess of...
-
Since when did military service become a right, for gays or anyone else? The military has always been allowed to discriminate against people who dont meet its requirements, including those who are...
-
we will be looking for:- four major types of planned change: human process interventions, techno-structural interventions, human resource management interventions, and strategic change interventions?...
-
Organizational Behavior Scholarly Journals only. Find an article that focuses on organizational behavior topics such as leadership, or decision making in your chosen field ( human resources. For...
-
Why would a leader demonstrating emotional intelligence be especially motivating to followers? How is self-monitoring related to emotional intelligence? How are these concepts (self-monitoring and...
-
Identify the components of an information system using the five-component framework and provide a brief summary of each. Explain Porter's five forces model. Management information systems...
-
Analyze how Filtronic used Arena to eliminate its time-consuming struggles with its internally developed product development tool that made tracking, maintaining, and releasing products slow and...
-
Selected data concerning the past years operations of the Ozarks Manufacturing Company are as follows: Required: 1. What was the cost of raw materials purchased during the year? 2. What was the...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Propose a mechanism that can explain the occurrence of this reaction: 0 CH2
-
When acetone is treated with anhydrous ammonia in the presence of anhydrous calcium chloride (a common drying agent), crystalline product C is obtained on concentration of the organic liquid phase of...
-
The difference in positive-charge distribution in an amide that accepts a proton on its oxygen or its nitrogen atom can be visualized with electrostatic potential maps. Consider the electrostatic...
-
Problem 2. Ground Tracks Revisited The ground tracks for three satellites (1,2,3) are shown in the figure. At time to the positions of the satellites in their orbits are indicated by the black...
-
Tasks: 1. Put unknown resistors R1 and R2 in series. Place this combination in parallel with resistor R3. Use the DMM as an Ohmmeter to measure the equivalent resistance of the circuit. [Setup...
-
4. Using the graphical polygon method, neatly construct a graphical solution for the three force vectors. Measure the resultant in magnitude and direction. Record your answer in Table III. Attach...

Study smarter with the SolutionInn App


