Analyze the aldol condensation in Eq. 22.53 using the method given in Eq. 22.54. Show that four
Question:
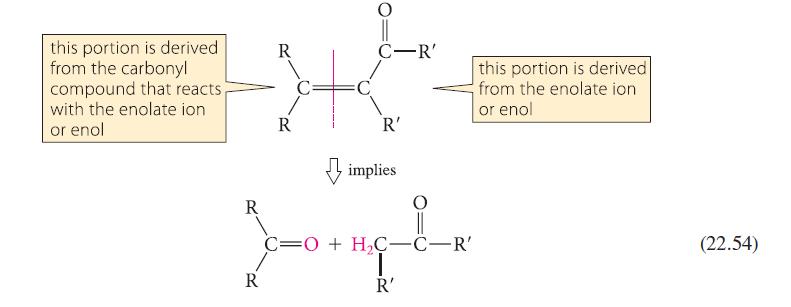
Analyze the aldol condensation in Eq. 22.53 using the method given in Eq. 22.54. Show that four possible aldol condensation products might in principle result from the starting material. Explain why the observed product is the most reasonable one.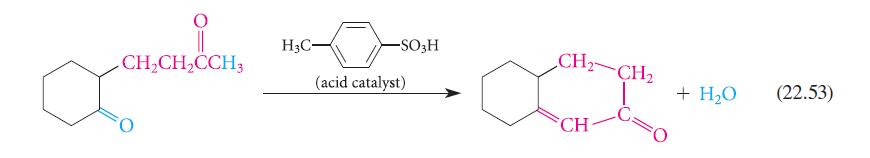
Transcribed Image Text:
CH₂CH₂CCH3 O H3C- -SO3H (acid catalyst) CH₂ CH₂ Ca CH + H₂O (22.53)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
First break the double bond as shown in Eq 2254 to reveal the possible starting material The ...View the full answer

Answered By

Emel Khan
I have the ability to effectively communicate and demonstrate concepts to students. Through my practical application of the subject required, I am able to provide real-world examples and clarify complex ideas. This helps students to better understand and retain the information, leading to improved performance and confidence in their abilities. Additionally, my hands-on approach allows for interactive lessons and personalized instruction, catering to the individual needs and learning styles of each student.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Q1. How worried are clients and stakeholders in day-to- day product improvement? 2. the industrial corporation Case for Agility "The struggle is not always to the most powerful, nor the race to the...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
A company that makes cola drinks states that the mean caffeine content per 12-ounce bottle of cola is 50 milligrams. You want to test this claim. During your tests, you find that a random sample of...
-
Mountain Cycles uses the FIFO inventory method. Mountain started August with 12 bicycles that cost $42 each. On August 16, Mountain bought 40 bicycles at $68 each. On August 31, Mountain sold 36...
-
Suppose you own 50,000 shares of common stock in a firm with 2.5 million total shares outstanding. The firm announces a plan to sell an additional 1 million shares through a rights offering. The...
-
Tyler Companys ASC 932-235 disclosures included the following information: REQUIRED: Using the information for Tyler Company in problems 13, 14, and 15 and in this problem: a. Compute the value of...
-
Sticky Polymers Inc. processes a base chemical into plastic. Standard costs and actual costs for direct materials, direct labor, and factory overhead incurred for the manufacture of 10,750 units of...
-
5.) A hoop of 6 kg and radius .15 m is rolling down a 40-degree incline with 5 m length, what acceleration does it travel down the incline? How long does it take to get down the incline?
-
The following reaction is known to involve enamine formation between the amine catalyst and the carbonyl marked with an asterisk (*). Draw a curved-arrow mechanism for this reaction, using the...
-
(a) The enzyme KDPG aldolase catalyzes the aldol addition reaction between pyruvate and glyceraldehyde-3-phosphate. The reaction is known to involve the formation of an imine (Schiff base) between a...
-
What is an inflection point? Give an example. What physical significance do inflection points sometimes have?
-
A- Small electricity market has a demand varies between 300 and 600 MW with a required 200 MW minimum spinning reserve. The table shows the characteristics of the available four generating units. ~ 3...
-
Find an equation of the plane perpendicular to D and through P, where D = 10i 10j + 5k and P is (1,1,-3). Find a plane crossing through (1,3,3), parallel to the plane 3x + y -z = 8. Is it possible to...
-
What role does data warehousing play in modern enterprise architectures, and how do techniques such as star schemas, snowflake schemas, and data cubes facilitate the transformation, aggregation, and...
-
Determine the value of X if any at which each function is discontinuous. at each number where F is this discontinuous state the conditions for continuity that are violated . f(x) = 2-1 x+1 if x # -1...
-
Prepare a trial balance Transaction Journal Entries 1 Paid monthly rent of $400. 2 Received $800 from customers when service was rendered. 3 Issued common stock to investors for $20,000. 4 Billed...
-
Explain why the quantity purchased (PQ) is used in computing the direct material price variance, but the actual quantity consumed (AQ) is used in computing the direct-material quantity variance.
-
A business had revenues of $280,000 and operating expenses of $315,000. Did the business (a) Incur a net loss (b) Realize net income?
-
Write a mechanism for the following reaction. (PhCO2)2, heat + CO
-
Hydrogen peroxide and ferrous sulfate react to produce hydroxyl radical (HO), as reported in 1894 by English chemist H. J. H. Fenton. When tert-butyl alcohol is treated with HO generated this way, it...
-
The halogen atom of an alkyl halide can be replaced by the hydrogen atom bonded to tin in tributyltin hydride (Bu3SnH). The process, called dehalogenation, is a radical reaction, and it can be...
-
4.6 Design a Lapple conventional cyclone to function as a precleaner. The particles have a density of 1250 kg/m, and have the same size distribution as in Problem 4.4, but the air is at 150 F and 1...
-
4.4 Recalculate the efficiency of the cyclone of Problem 4.2 for the fol- lowing particle size distribution. Size Range, um 0-2 Mass Percent in Size Range 4.0 2-6 16.0 6-12 20.0 12-20 20.0 20-40 20.0...
-
Question 2 (a) Two wide plates are fabricated from a 4340 alloy steel. Each of these plates is then subjected to a different heat treatment to yield a different set of mechanical properties. Table...

Study smarter with the SolutionInn App


