Arrange the following four alkyl halides in descending order with respect to the E2 elimination to S
Question:
Arrange the following four alkyl halides in descending order with respect to the E2 elimination to SN2 substitution product ratio expected in their reactions with sodium ethoxide in ethyl alcohol. Explain your answers.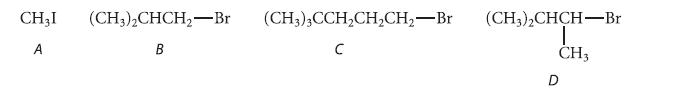
Transcribed Image Text:
CH,I (CH3)2CHCH₂-Br A B (CH3)3CCH₂CH₂CH₂-Br C (CH3)2CHCH-Br I CH3 D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Analyze the alkyl substitution pattern in each alkyl halide alkyl substitution at both the ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Arrange the following four bases in descending order with respect to the E2 elimination to S N 2 substitution product ratio expected when they react with isobutyl bromide. Explain your answers....
-
Yasser has just won the Quebec Lottery with a jackpot of $1,000,000. His winnings will be paid out in 50 equal annual installments with the first payment made immediately! He believes the appropriate...
-
Write a parallel MPI application that finds 2-grams (bigram) in the news dataset (35 MB compressed) in the shortest time. (You can find the compressed file here...
-
Mr. Lawrence had been the manager at Pleasure Sdn Bhd (PSB) since 1 February 2016. His employment was terminated on 30 April 2021 due to disputes with the directors of PSB. After many appeals,...
-
Full Disclosure Principle Presented below are a number of facts related to Weller, Inc. Assume that no mention of these facts was made in the financial statements and the related notes. Instructions...
-
Suppose the trade imbalances of the 2000s largely disappear during the next decade. What is likely to happen to the huge global capital flows of the 2000s? What is the link between the trade...
-
Consider the net cash flows and salvage values shown below. Assume the alternatives can be indefinitely renewed with the same cash flows and salvage values. Using a MARR of 8%, specify the planning...
-
Liability Entries and Adjustments Listed below are selected transactions of Schultz Department Store for the current year ending December 31. 1. On December 5, the store received $500 from the...
-
Direct Materials Conversion Percent Percent Units Complete Beginning work in process inventory 2,800 100% Complete 25% Units started this period 6,800 Completed and transferred out 7,800 Ending work...
-
Write a curved-arrow mechanism for formation of the rearrangement product shown in Eq. 9.60. CH3 CH3 I T H3C-C- CH 3 -CH-Cl EtOH 80 C CH3 CH3 | I H3C-C- | OEt + -CH-CH3 EtOH Cl + other products...
-
What nucleophile or base and what type of solvent could be used for the conversion of isobutyl bromide into each of the following compounds? (a) (CH3)CHCHS (CH3)2 Br (b) (CH3)CHCHSCHCH3 (c)...
-
(a) If accounts payable are 40,000 and purchases are 240,000, what is the accounts payable/purchases ratio? (b) How many days does the business take on average to pay its creditors?
-
You have decided to buy a perpetuity. The bond makes one payment at the end of every year forever and has an interest rate of 4%. If you initially put $5000 into the bond, what is the payment every...
-
Why does an FSNC typically have several aircraft types in its fleet?
-
How important are the following: (a) the grapevine; (b) nonverbal communication?
-
Comment on the signifcance of cultural diferences in communication.
-
What are the characteristics of state- owned airlines that often render them uncompetitive with privately (or public stock) owned airlines.
-
A company purchases an asset that costs $10,000. This asset qualifies as three-year property under MACRS. The company uses an after-tax discount rate of 12 percent and faces a 40 percent income-tax...
-
Refer to the Conservation Ecology (Dec. 2003) study of the causes of forest fragmentation, presented in Exercise 2.166 (p. 97). Recall that the researchers used advanced high-resolution satellite...
-
Give IUPAC names for the following alkylhalides: (b) (a) CHCH2CH2CH21 H C (c) C,2I BrCH2CH2CH2CH2Br CH (e) Br (1) 1 -CH2CI CI CH (d) CH2H2CI CHH2H CHCH2CH2CH I
-
Draw structures corresponding to the following IUPAC names: (a) 2-Chloro-3, 3-dimethylhexane (b) 3, 3-Dichloro-2-methylhexane (c) 3-Bromo-3-ethylpentane (d) 1, 1-Dibromo-4-isopropylcyclohexane (e)...
-
Draw and name all monochloro products you would expect to obtain from radical chlorination of 2-methylpentane. Which, if any, are chiral?
-
How does trade allow for specialization? How does trade result in greater overall output:? What is the difference between comparative advantage and absolute advantage? What are the...
-
In the US, the market for Ibuprofen is competitive with a large number of producers and consumers. Ibuprofen faces a lot of competition, in part, because there are many available substitutes, such as...
-
Directions: First, respond to the question below; Second, compose a multi-paragraph essay that has an average length of two hand-written pages; Third, plan to spend about one hour on the essay, being...

Study smarter with the SolutionInn App


