At 368C the NMR resonances for the ring methyl groups of isopropylmesitylene (protons H a and H
Question:
At 36°8C the NMR resonances for the ring methyl groups of “isopropylmesitylene” (protons Ha and Hb in the following structure) are two singlets at δ 2.25 and δ 2.13 with a 2 : 1 intensity ratio, respectively. When the spectrum is taken at 260°8C, however, it shows three singlets of equal intensity for these groups at δ 2.25, δ 2.17, and δ 2.11. Explain these results.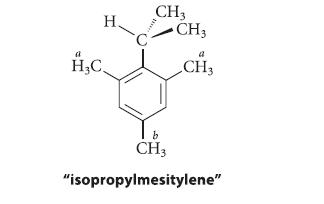
Transcribed Image Text:
H H₂C CH3 CH3 a CH3 CH3 "isopropylmesitylene"
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
At the higher temperature the two ortho methyl groups labeled a i...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Influencer marketing includes only movie, tv and sports celebrities with significant social media audiences. True False 4
-
Histogram. Suppose that the standard input stream is a sequence of double values. Write a program that takes an integer n and two real numbers lo and hi as command-line arguments and uses StdDraw to...
-
At December 31, 2011, Ethan Company Reports the following results for its calendar-year. Cash Sales $1,803,750 Credit Sales 3,534,000 In addition, its unadjusted trail balance includes the following...
-
If conflicts develop into a situation where mistrust prevails, would you expect activity documentation to increase or decrease? Why?
-
A \(20 \Omega\) resistor is connected across a \(120 \mathrm{~V}\) source. The resistor is then lowered into an insulated beaker, containing \(1.0 \mathrm{~L}\) of water at \(20^{\circ} \mathrm{C}\),...
-
Use Worksheet 9.2. Bruce Kaplan, a 35-year-old computer programmer, earns $72,000 a year. His monthly take-home pay is $3,750. His wife, Barbara, works part-time at their childrens elementary school...
-
You are a manager at Northern Fibre, which is considering expanding its operations in synthetic fibre manufacturing. Your boss comes into your office, drops a consultant's report on your desk, and...
-
A farmer can buy four types of plant food. Each barrel of mix A contains 30 pounds of phosphoric acid, 50 pounds of nitrogen, and 30 pounds of potash; each barrel of mix B contains 30 pounds of...
-
Draw a variation on the rebound hydroxylation mechanism that accounts for the formation of epoxides from some alkenes by CyP450 as shown in Fig. P17.38. C=C +47 +47 Felv Fell S S. Figure P17.38...
-
Information for Ditka Inc. and McMahon Company is given below: Required: 1. Compute Ditka's and McMahon's free cash flow and cash flow adequacy ratio. 2. What information do these cash-based...
-
If an employee is covered by a group-term life insurance plan that pays the entire premium of the policy, must the employee include the premium coverage in income if the face value of the policy is...
-
Which relative does not have to live in the same household of the taxpayer claiming head of household filing status? a. Aunt b. Son c. Granddaughter d. Father e. Brother
-
What filing status options are available to Darlene, assuming she separated from her husband in March? Darlene is the sole support of her daughter, who lives with Darlene.
-
Which of the following is not considered when determining the total support test for a child? a. Fair rental value of lodging b. Medical insurance premiums c. Birthday presents d. Scholarship e....
-
How do head of household expenses differ from the expenses used in the support test?
-
Use Tukey's HSD test to compute multiple comparisons for the data in problem 11.12. Let = .01. State which regions, if any, are significantly different from other regions in mean starting salary...
-
What kind of rays are X-rays?
-
Complete the following reactions. If no reaction is tikety, explain why. (a) (b) CH,SH + NaOH -_, (1 equiv.) 25 C CH OH
-
Outline a synthesis of each ether using either alcohol dehydration or alkene addition, as appropriate. (a) 2-methoxy-2-methylbutane (b) dibutyl ether
-
Give the structure of the that would with mCPBA to give each of the following expoxides. (a) (b) . /A C CH2 H,C C-4 CH,
-
Write a program in MATLAB to find the average of three numbers.
-
If $ 1 3 , 3 7 6 . 3 9 was the interest earned on lending $ 1 0 1 , 4 0 0 . 0 0 for 4 years, what was the quarterly compounding nominal interest rate charged on the loan?
-
A long forward contract on a non - dividend - paying stock was entered into some time ago. It currently has six months to maturity. The risk - free rate of interest ( with continuous compounding ) is...

Study smarter with the SolutionInn App


