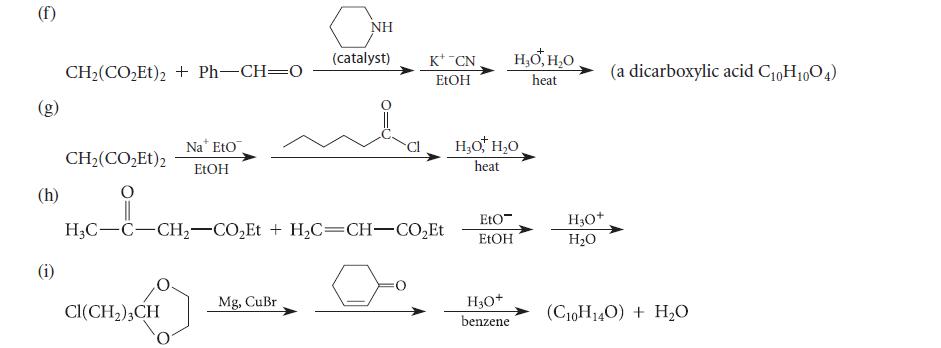
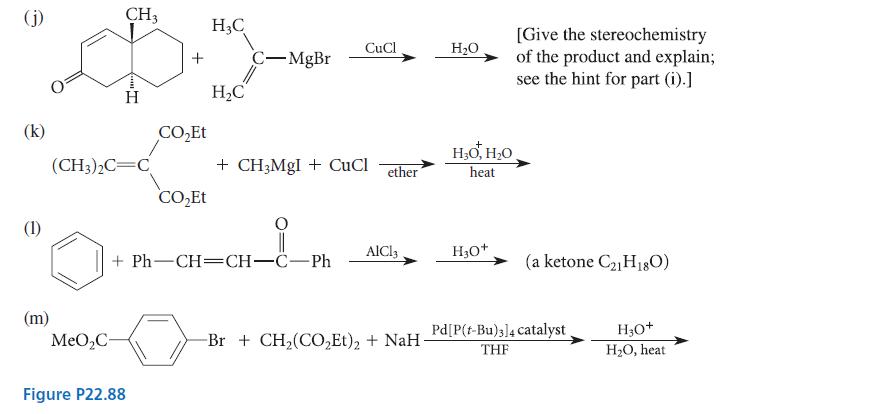
Complete the reactions given in Fig. P22.88 by giving the major organic products. Explain your reasoning. (b)
Question:
Complete the reactions given in Fig. P22.88 by giving the major organic products. Explain your reasoning.
![(b) (c) (d) (e) 1-CH-i H3C-C-CH-C-OEt + Br(CH)4Br & y-butyrolactone Cl NO CH3 NO Li* [(CH3)zCH],N: +Na+](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/8/6/4/5986570649613da71701864595336.jpg)
Transcribed Image Text:
Ⓡ (b) (c) (d) (e) 1-CH₂-i H3C-C-CH₂-C-OEt + Br(CH₂)4Br & y-butyrolactone Cl NO₂ CH3 NO₂ Li* [(CH3)zCH],N: +Na+ :CH(CO₂Et) 2 C-CH3 1) LiAlH4 2) H3O+ CH 31 NaOEt -CO₂Et + CH3CH(CO₂Et)2 EtOH NaOEt (excess) EtOH H3O+ H₂O heat H3O+ heat (C₂H12O)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The conjugatebase enolate ion is alkylated by the alkyl halide Then the conjugatebase enolate ion of the alkylation product is formed and reacts int...View the full answer

Answered By

Mishark muli
Having any assignments and any other research related work? worry less for I am ready to help you with any task. I am quality oriented and dedicated always to produce good and presentable work for the client once he/she entrusts me with their work. i guarantee also non plagiarized work and well researched work to give you straight As in all your units.Feel free to consult me for any help and you will never regret
4.70+
11+ Reviews
37+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
Complete the following reactions by giving the major organic products. (a) (b) (c) (d) (CH)NH (excess) SOCI CH,CH.CO,H-(excess) Cl-C-CI CH OH (excess O0 O + CH,OH-
-
Complete the reactions given in Fig. P19.45 by giving the principal organic product(s). Fig. P19.45 (a) (b) (c) p-toluenesulfonic acid (catalyst CH, t CH,OH - (solvent) ether Hio -caphenone, +...
-
Suppose a country's productivity last year was 84. If this country's productivity growth rate of 5 percent is to be maintained, this means that this year's productivity will have to be: A. 88.2. B....
-
What is the difference between an account receivable and a note receivable?
-
Kampa Company and Arbor Company are similar firms that operate in the same industry. Arbor began operations in 2001 and Kampa in 1995. In 2006, both companies pay 7% interest on their debt to...
-
Describe the role of an organizations personnel in compliance and antifraud efforts.
-
Highland Company produces a lightweight backpack that is popular with college students. Standard variable costs relating to a single backpack are given below: Overhead is applied to production on the...
-
Consider graph G below. We want to construct a minimum spanning tree out of that. In what order are edges will be added to the Minimum Spanning Tree (MST) using Kruskal's Algorithm? Order is from...
-
When the epoxide 2-vinyloxirane reacts with lithium dibutylcuprate, followed by protonolysis, a compound A is the major product formed. Oxidation of A with PCC yields B, a compound that gives a...
-
Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.87. H3C C-CH3 (c) (b) BrCHCO,CH3 I (CH)3 T BrCHCO,CH3 2 COEt KCO3 COEt NaOEt + 2 NaH HO CH3 DMF HO,...
-
Beowulf and Grendel, a public accounting firm in London, is engaged in the preparation of income tax returns for individuals. The firm uses the weighted-average method of process costing for internal...
-
As the SA Human Rights Commission (SAHRC) is set to convene an inquiry into the advertising industry and its adherence to human rights, Unilever says it was "shocked to discover" the material they...
-
According to Quintanilla and Wahl (2019), team members share leadership responsibilities. Team leaders help to facilitate communication and productivity during meetings. the following article from...
-
In 2021 Boiler Organization paid $35000 to Purdue Products for a piece of land owned by Purdue Products. The land (which has an adjusted basis of $19000) will be used as a site for Boiler's new...
-
A new Latina graduate of an MHSA program is hired to be in charge of a quality improvement and patient safety initiative at a hospital in Washington, DC. On her first morning at work, she asks her...
-
The actor Wesley Snipes was indicted in 2006 for not filing tax returns for the years 1999 through 2004 and filing refund claims for approximately $12 million for 1996 and 1997, claiming that only...
-
Kenneth Washburn, head of the Sporting Goods Division of Reliable Products, has just completed a miserable nine months. If it could have gone wrong, it did. Sales are down, income is down,...
-
The area of a rectangle is 30 cm 2 and its perimeter is 26 cm. Find the length and width of the rectangle.
-
Write structural formulas for the products that form when 1-butene reacts with each of the following reagents: (a) HI (b) H2, Pt (c) Dilute H2SO4, warm (d) Cold concentrated H2SO4 (e) Cold...
-
Repeat Exercise 8.26 using 1-methylcyclopentene instead of 1-butene. In Exercise 8.26 (a) HI (b) H2, Pt (c) Dilute H2SO4, warm (d) Cold concentrated H2SO4 (e) Cold concentrated H2SO4, then H2O and...
-
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable. (a) (b) (c) (d) (e) H20, H.So4 dr (1) BH THF (2) NaOH, H20, Br (1) 03 (2) Meps
-
A project management plan is the foundation for all projects and contains the key information requested in a typical funding application. Developing a project management plan is a key responsibility...
-
describe the possible defences available to a defendaant when a negligece action is brought against them, explain your answer in details
-
A thin disk of radius 1.2m has a uniform surface charge density = 0.0030 C/m. (a) (2 points) What is the total charge on the disk? (b) (5 points) You are asked to calculate the electric field at...

Study smarter with the SolutionInn App


