Complete the reactions shown in Fig. P24.45 by giving the major organic product(s). (a) (b) (e) phenyl
Question:
Complete the reactions shown in Fig. P24.45 by giving the major organic product(s).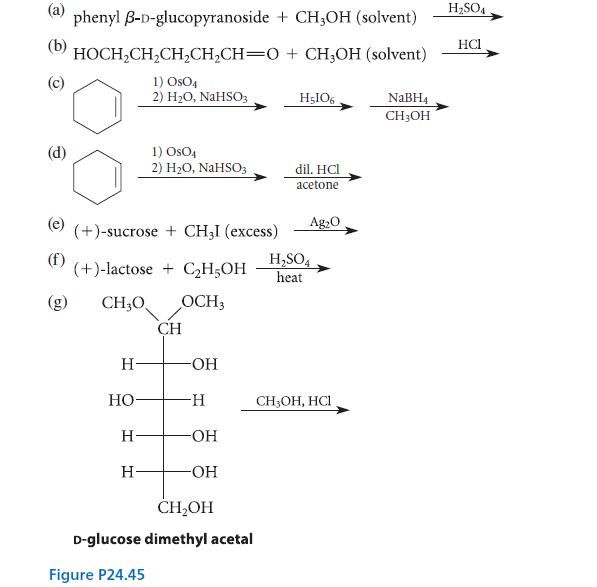
Transcribed Image Text:
(a) (b) (e) phenyl B-D-glucopyranoside + CH3OH (solvent) HOCH₂CH₂CH₂CH₂CH=O + CH3OH (solvent) 1) OsO4 2) H₂O, NaHSO3 (+)-sucrose + CH3I (excess) (+)-lactose + C₂H5OH CH₂O OCH3 H -OH -H -OH -OH CH₂OH D-glucose dimethyl acetal HO H 1) OSO4 2) H₂O, NaHSO3 H Figure P24.45 CH H5106 dil. HCI acetone H₂SO4 heat Ag₂O CH,OH, HCI NaBH4 CH3OH H₂SO4 HC1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a Just as the acetal units in carbohydrates undergo hydrolysis in water they also undergo methanolys...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete the following reactions by giving the major organic products. (a) (b) (c) (d) (CH)NH (excess) SOCI CH,CH.CO,H-(excess) Cl-C-CI CH OH (excess O0 O + CH,OH-
-
Complete the following reactions by giving the major organic product(s), including stereochemistry. (a) Ph-C=C-Ph+ (b) (c) CH3 0- CH CH3 CH3 + HC HC O C=C CH3 CH3 heat heat light (C20H22)
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
IFRS requires the following: (a) Ending inventory is written up and down to net realizable value each reporting period. (b) Ending inventory is written down to net realizable value but cannot be...
-
Homer enters the visitors chambers, and his geiger-beeper goes off. He shuts off the beep, removes the device from his shoulder patch and holds it near the only new object in the room: an orb which...
-
Raymond Manufacturing faces a liquidity crisisit needs a loan of $100,000 for 1 month. Having no source of additional unsecured borrowing, the firm must find a secured short-term lender. The firms...
-
Prove that, if \(\tau\) is an initial time with \(\mathbb{E}_{\mathbb{Q}}\left(1 / \alpha_{\infty}^{\tau} ight)
-
The following is a summary of all relevant transactions of Vicario Corporation since it was organized in 2012. In 2012, 15,000 shares were authorized and 7,000 shares of common stock ($50 par value)...
-
Calculate the decimal equivalents of the following binary numbers: 1. 11011011 2. 01111111 3. 10000000 4. 11000000 5. 11001101
-
Chlorotris(triphenylphosphine)rhodium brings about the decarbonylation of aldehydes: (a) What product is obtained when this reaction is applied to D-galactose? (b) Suggest a reason why the reaction...
-
An important reaction used by Emil Fischer in his research on carbohydrate chemistry was the reaction of aldoses and ketoses with phenylhydrazine to give osazones, shown in Fig. P24.44. Osazones,...
-
Dr. Paul Siple conducted studies testing the effect of wind on the formation of ice at various temperatures and developed the concept of wind chill, which we hear reported during winter weather...
-
Consider your surroundings; what are the products that you could acquire as a service instead of buying, owning, and trashing them?
-
How might women discern between traditional stereotype-based roles and opportunities that benefit from dual agentic and communal expectations?
-
How might higher education leaders more intentionally and authentically harness their institutional history and heritage to respond to the VUCA of our contemporary context?
-
How are managers equipped to manage the change process in terms of cultivating servant leadership behaviors such as employee empowerment?
-
What is self-leadership?
-
In a recent year, the average daily circulation of The Wall Street Journal was 1,717,000. Suppose the standard deviation is 50,940. Assume the paper's daily circulation is normally distributed. On...
-
For the data in Exercise 17-19, use the FIFO method to summarize total costs to account for, and assign these costs to units completed and transferred out, and to units in ending work in process....
-
Using the curved-arrow notation, fill in the details for base-catalyzed mutarotation of glucopyranose. Begin by remwing a proton from the hydroxy group at carbon-1.
-
By drawing resonance structures for the carbocation intermediates, show why aromatic substitution in pyridine - N - oxide occurs at the 4 - position rather than at the 3 - position.
-
Which compound should undergo substitution of the bromine by phenolate anion: 4 - bromopyridine or 3 - bromopyridine ? Explain, and give the structure of the product.
-
4. APPLICATION The case study indicates that the City of Lakeview will not have any additional debt capacity two years from now. The additional debt service ratio is negative (-1.10 percent),...
-
What are the key challenges will be faced by business like Flipkart in future? State your view with suitable example.
-
Why volume based discount is preferred? Explain your answer with suitable example.

Study smarter with the SolutionInn App


