Consider the reaction of a methyl radical (CH3) with the p bond of an alkene: The relative
Question:
Consider the reaction of a methyl radical (·CH3) with the p bond of an alkene: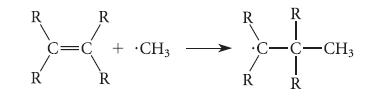
The relative rates of the reaction shown in Fig. P5.50 were determined for various alkenes.
(a) Draw the free-radical product of the reaction in each case and explain.
(b) Explain the order of the relative rates.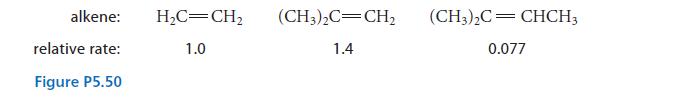
Transcribed Image Text:
R R T Hoa C=C + CH3 R R R R R R •C-C-CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a The methyl radical reacts at the carbon of the double bond with fewer substituents because this mode of reaction gives the more substituted radical ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction of a methyl ketone with iodine solution in the presence of aqueous sodium hydroxide is shown in the equation below: RCOCH3() + 3l2(aq) + 4NaOH(aq) RCOONa(aq) + CHl3(s) + 3Nal(aq) +...
-
Namib Mills, has a fixed total capital of $10,000,000, which is made up of 20 percent debt and 80 percent equity. The firm has 100,000 outstanding ordinary shares and no preference shares. Although...
-
Suppose an industry has the following data: TC=q-5q+100 a P=55-2Q If the industry is competitive; Calculate price, production quantity and profit. b. If there is a firm in the industry; Calculate...
-
Think about the impact that e-commerce and more flexible networks of organisations has had on the way international business is conducted. How has it created new complexities in the relationships...
-
H. Banks Company would like to design, produce, and sell versatile toasters for the home kitchen market. The toaster will have four slots that adjust in thickness to accommodate both slim slices of...
-
What are the differences between total consideration, total purchase price/enterprise value, and net purchase price? How are these different concepts used?
-
Use information from Section 6.7 to estimate which form of electromagnetic radiation is the lowest energy ionizing radiation. Data from section 6.7 When we first introduced the concept of the...
-
Breyer Company purchased packaging equipment on January 3, 2010, for $101,250. The equipment was expected to have a useful life of three years, or 25,000 operating hours, and a residual value of...
-
Map the EER model of the following databases, to a relational model. Show all integrity types(Clarify your answer by underline the Primary key, and dotted line the Foreign Key). (a) International...
-
Equations 5.25ac on p. 193 show the formation of trialkylboranes from alkenes and BH 3 . In the reaction of 2,3-dimethyl-2-butene with BH 3 , only two equivalents of the alkene react, even with a...
-
Using the curved-arrow or fishhook notation, as appropriate, suggest mechanisms for each of the reactions given in Fig. P5.49. (a) HC (b) H3C C-CH CHCH=CH HC CH T T HO OH C#yan + Br H3O+ HC + CI-S-CI...
-
The following pension plan information was assembled by the finance director for Dungannon County for the fiscal year ended June 30, 20X6, which is also the measurement date for the net pension...
-
What is the current opportunity cost of a five-year old concrete wall around a property if the wall originally cost 500,000 to build, is depreciated straight-line over 20 years, and the owner has a...
-
George is 37 years old and divorced. He has a seven-year-old son, Joshua, whom he claims as a dependent. George's gross income was $17,000 during the tax year. Based on this information, is he...
-
3. Consider the following loop. LOOP: LDUR X10, [X1, #0] LDUR X11, [X1, #8] ADD X12, X10, X11 SUBI X1, X1, #16 CBNZ X12, LOOP (10 points) Assume that perfect branch prediction is used (no stalls due...
-
Each of the three STR loci used in this analysis assort independently of each other during meiosis. Therefore, the inheritance of the alleles at each STR locus is mutually exclusive to another STR...
-
At a certain college,45%of the students are female, and17%of the students major in civil engineering. Furthermore,10%of the students both are female and major in civil engineering. (a)What is the...
-
Refer to the financial statements and related disclosure notes of Dell Inc. located in the company's annual report for the fiscal year ending January 30, 2009, included with all new copies of the...
-
Prepare a stock card using the following information A company is registered for GST which it pays quarterly, assume GST was last paid on the 30th of June 2019. It uses weighted average cost...
-
The energy level diagram for the MOs of CH 2 = CH 2 is shown if Figure 3.24. Show a similar diagram for the lowest-energy excited state of this molecule. C=C 5 o* * Ethene 5 o Energy
-
Consider the species formed by the addition of an extra electron to H2 so that there are three electrons and a negative charge. Show an energy level diagram for the MOs of this species. Is there...
-
Draw an energy level diagram for the excited state of H2. Is there still a bond between the hydrogens?
-
What is the major premise behind activity-based costing? please explain.
-
Now, let's consider the AD/AS model to understand the impact of the zero COVID-19 policy and the subsequent fiscal policy measures: The term "fiscal policy" is used to describe the government's...
-
Uber charges a price per mile of $4. they are considering a game day surge price strategy. Estimated demand functions : Group 1 rides before game Q1=500-100P1 Group 2 riders after the game...

Study smarter with the SolutionInn App


