Decide whether each of the following compounds is aromatic. Explain your reasoning. (a) (d) -CH3 toluene (b)
Question:
Decide whether each of the following compounds is aromatic. Explain your reasoning.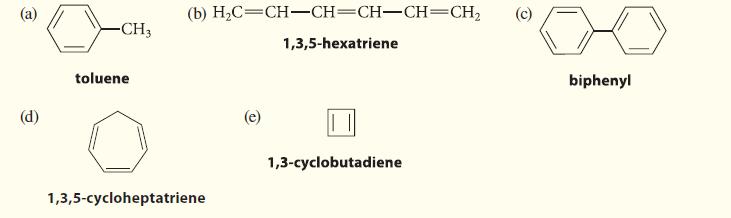
Transcribed Image Text:
(a) (d) -CH3 toluene (b) H₂C=CH-CH=CH-CH=CH₂ 1,3,5-hexatriene 1,3,5-cycloheptatriene (e) 1,3-cyclobutadiene (c) biphenyl
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
In each example first count the electrons by applying the following rule Each double bond contribute...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following compounds is an aromatic compound bearing a substituent that we did not discuss in this chapter. Using the principles that we discussed in this chapter, predict the major...
-
Determine whether each of the following compounds is a cis isomer or a trans isomer a. b. c. d. e. f. Cl Br CH3 CH3 Br Br CH3 Cl CH3 CH3 CH
-
Predict whether each of the following compounds is likely to be dangerously explosive in contact with BrF 3 and explain your answer: (a) SbF 5 , (b) CH 3 OH, (c) F 2 , (d) S 2 Cl 2 .
-
discuss the benefits and challenges of the employee job performance evaluation process as it relates to the ratings of an individual. For example, if an employer uses a standard Likert Scale (1...
-
Give an example of an asset exchange transaction. What is the effect of this transaction on the accounting equation?
-
Bowdeen Manufacturing intends to issue callable, perpetual bonds with annual coupon payments. The bonds are callable at $1,175. One-year interest rates are 9 percent. There is a 60 percent...
-
Tom Bonacci brought his Jeep to Brewer Service Station to investigate a strange noise the vehicle was making. The Jeep was raised up on an automobile lift so that Brewer employee Paul Gebing could...
-
Little Theatre is a nonprofit organization devoted to staging plays for children. The theater has a very small full-time professional administrative staff. Through a special arrangement with the...
-
The storied American aerospace company, McDonnell Douglas, designed a wide-bodied airliner in the mid-1980s called the MD-11. Research and development costs, plus other fixed costs related to simply...
-
Explain why there is a larger difference between the heats of formation of (E)-1,3 - pentadiene and 1,4-pentadiene (29.3 kJ mol 1 or 7.1 kcal mol 1 ) than between (E)-1,3- hexadiene and...
-
Use the Frost circle to determine the energy levels and electron occupancies for the MOs of benzene.
-
A laser beam illuminates a single, narrow slit, and the diffraction pattern is observed on a screen behind the slit. The first secondary maximum is 26 mm from the center of the diffraction pattern....
-
Which methods are most used in practice?
-
When quantity supplied is greater than quantity demanded, ___________-. a) price will fall to its equilibrium level b) price will rise to its equilibrium level c) price may rise, fall, or stay the...
-
Provide a list of pros and cons of test marketing.
-
How might an automobile company use ethnography to improve their product?
-
Explain the two types of validity in experimentation. What types of experiments are best suited to address each?
-
Abe is an employee of Haddock, Inc. Haddock provides basic health and accident insurance to all its employees through a contract with Minor Accident Insurance Company. Because the Minor policy does...
-
You are a Loan Officer with an Investment Bank. Today you need to set your lending parameters. They are: LTV: 55% 10 Year T-Bill: TBD Rate Markup: 300 Basis Points Term: 30 Years Amortization: 30...
-
Propose a structure for compound C, which has M + = 86 in its mass spectrum, an IR absorption at 3400 cm 1 , and the following 13 C NMR spectral data: Compound C Broadband-decoupled 13 C NMR: 30.2,...
-
Compound D is isomeric with compound C (Problem 13.61) and has the following 13C NMR spectral data. Propose a structure. Compound D Broadband-decoupled 13C NMR: 9.7, 29.9, 74.4, 114.4, 141.4 DEPT-90:...
-
Propose a structure for compound E, C7H12O2, which has the following 13C NMR spectral data: Compound E Broadband-decoupled 13C NMR: 19.1, 28.0, 70.5, 129.0, 129.8, 165.8 DEPT-90: 28.0, 129.8 ...
-
A 100,000.00 mortgage has a 25-year amortization, at 3% interest compounded annually with a monthly mortgage payment. a. How much are the monthly mortgage payments? (2 points) b. After 5 years (60...
-
What is a mathematical formula to calculate Inventory as a Percentage of Total Inventory?
-
In a business setting, daily incidental expenses are managed through petty cash. According to accounting principles, where should petty cash disbursements for these expenses be recorded on a daily...

Study smarter with the SolutionInn App


