Design a synthesis of methyl orange (Eq. 23.49) using aniline as the only aromatic starting material. -N=N-
Question:
Design a synthesis of methyl orange (Eq. 23.49) using aniline as the only aromatic starting material.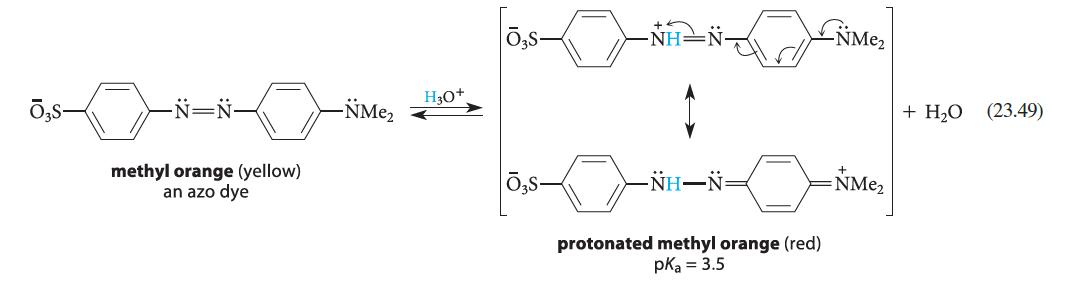
Transcribed Image Text:
-N=N- methyl orange (yellow) an azo dye -NMe₂ H3O+ NH–N protonated methyl orange (red) pka = 3.5 NME₂ =NMe₂ + H₂O (23.49)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
pAminobenzenesulfonic acid is prepared and diazotized to give the diazonium ion A This syn...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the flow rate in each tube of the system shown. The pipes are cast iron; the flow rate in the shunts is expressed in liters/sec. 9.5 159.3 305 m 458m 9.5 11 203mm 458m 305m 254mm 203mm 153m...
-
Using benzene or toluene as the only aromatic organic starting material, devise a synthesis for each of the following compounds. Name the product. CH CH3 Cl CH3 Cl a. b. C. d. Br NO2 NO2
-
Design a synthesis of 3-pentyn-1-ol using propyne and ethylene oxide as the only sources of carbon atoms.
-
Abhishek Ltd. is manufacturing cotton clothes. It has been consistently earning good profits for many years. This year too, it has been able to generate enough profits. There is availability of...
-
Match the following users with the information needed. 1. Owners 2. Managers 3. Creditors 4. Government agencies a. Whether the firm can pay its bills on time b. Detailed, up-to-date information to...
-
What account balances within the current year might be used for analytical procedures? How might balances between years be used for analytical procedures?
-
True or False: For personal investment decision making, rates of return are used more frequently than present worth.
-
Recognition of Profit on Long-Term Contracts During 2010 Nilsen Company started a construction job with a contract price of $1,600,000. The job was completed in 2012. The following information is...
-
A pre-anoxic MLE activated sludge process has the following influent wastewater characteristics and is operated with an anoxic volume of 500 m3 and MLVSS of 2000 mg/L at 20 oC. Given that influent...
-
(a) Which one of the following three amines can be prepared by either the Gabriel synthesis or the Staudinger reaction: 2,2-dimethyl-1-propanamine, 3-methyl-1-pentanamine, or N-butylaniline? (b)...
-
(a) Write a Lewis structure for HNO 2 in Eq. 23.50. (b) Write a mechanism for the reaction shown in Eq. 23.50. Me,NH + HNO, + HO MeN N,N-dimethylnitrosamine (89-90% yield) (23.50)
-
Instantaneous Power in a Standing Wave. From Eq. (15.21), the instantaneous rate at which a wave transmits energy along a string (instantaneous power) is Where F is the tension. (a) Evaluate f (x, t}...
-
A taxpayer wishes to buy some electric vehicles for his business and get the Commerical Clean Vehicle Credit for his purchases when he files for TY2023. He plans on buying four (4) vehicles, only one...
-
Consumption under borrowing constraint (a) With borrowing constraint, household can borrow until 200 in period 1. Under y1 =100, y2= 200, r= 0.2, How much is maximum possible consumption of period 1?...
-
In each of Problems 1 through 4, determine "(xo), " (xo), and (4)(xo) for the given point to if y = o(x) is a solution of the given initial value problem. 1. y + xy + y = 0; y(0) = 1, 2. y + (sin...
-
A taxpayer operating a sole proprietorship and filing on a Schedule C pays $65,000 in health insurance premiums for himself and his employees. $15,000 of that amount is for premiums paid to cover...
-
4. Given the following for a carbide insert single point tool, what is the expected tool life for a high-speed operation given: . = 257.35 b. Vc = 10 m/sec c. So = 1mm/rev d. T = 3mm e. y = 0.2...
-
Why should the system request be created by a businessperson as opposed to an IS professional?
-
The Ferris wheel in the figure has a radius of 68 feet. The clearance between the wheel and the ground is 14 feet. The rectangular coordinate system shown has its origin on the ground directly below...
-
Draw a hypothetical free-energy diagram for the SN2 reaction of iodide anion with 1-chlorobutane. Label the diagram as in Fig. 6.4, and assume it is exergonic but without specific values for G¡...
-
When ethyl bromide reacts with potassium cyanide in methanol, the major product is CH3CH2CN. Some CH3CH2NC is formed as well, however. Write Lewis structures for the cyanide ion and for both products...
-
Give structures for the products of each of the following reactions: (a) (b) (c) (d) (e) Nal (1 mol) H -acetone GH,H + NaBr Nal (1 mol), CH,NaCl CI CI BrBr (1 mol C4HS2 2 NaBr OH NaH (-H2) Cl Et O...
-
Can an economy be defined as how a country produces, distributes, and consumes valuable things?
-
Ecosolutions income statement is presented below: Sales Contribution Margin Income Statement Per Unit Total $30 $24,000,000 Variable Costs Direct Materials Direct Labor 4 3,200,000 5 4,000,000...
-
EE printing is in the evaluation process for the acquisition of a new computer system. The total depreciable base (cost plus installation) is $175,000. The new equipment will increase earnings before...

Study smarter with the SolutionInn App


