Explain how the compounds within each set can be distinguished using only UV spectroscopy. (a) 2-cyclohexenone and
Question:
Explain how the compounds within each set can be distinguished using only UV spectroscopy. 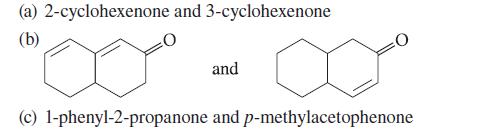
Transcribed Image Text:
(a) 2-cyclohexenone and 3-cyclohexenone (b) and (c) 1-phenyl-2-propanone and p-methylacetophenone
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a b c The double bonds in ...View the full answer

Answered By

Benish Ahmad
I'm a professional software engineer. I'm lectutrer at GCUF and I have 3 years of teaching experience. I'm looking forward to getting mostly computer science work including:
Programming fundamentals
Object oriented programming
Data structures
object oriented design and analysis
Database system
Computer networks
Discrete mathematics
Web application
I am expert in different computer languages such as C++, java, JavaScript, Sql, CSS, Python and C#. I'm also have excellent knowledge of essay writing and research. I have worked in other Freelancing website such as Fiverr and Upwork. Now I have finally decided to join the SolutionInn platform to continue with my explicit work of helping dear clients and students to achieve their academic dreams. I deliver plagiarism free work and exceptional projects on time. I am capable of working under high pressure.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain how the compounds within each set can be distinguished using only UV spectroscopy. 2-cyclohexenone and 3-cyclohexenone
-
Assume you have unlabeled samples of the compounds within each of the following sets. Explain how UV-vis spectroscopy could be used to distinguish each compound in the set from the other (s). (a) (b)...
-
Explain how you would distinguish the compounds within each set by a simple chemical test with readily observable results, such as solubility in acid or base, evolution of a gas, and so forth....
-
Tom Lamont, age 30, and Lin Lamont, age 31, have been married for six years. They got married right after Tom graduated from college. They have come to you for help in planning their financial...
-
Laughter Landscaping has the following independent cases at the end of the year on December 31, 2014. a. Each Friday, Laughter pays employees for the current weeks work. The amount of the weekly...
-
Let X be normally distributed with mean = 2.5 and standard deviation = 2> a. Find P(X>7.6)> b. Find P(7.4 X 10.6)> c. Find x such that P(X>x) = 0.025> d. Find x such that P(x X 2.5) = 0.4943>
-
A university pumps its water from wells located on campus. The falling water table has caused pumping costs to increase, the quantity of water available to decrease, and the quality of water to...
-
Flexible budget planning Luke Chou, the president of Digitech Computer Services, needs your help. He wonders about the potential effects on the firm's net income if he changes the service rate that...
-
Transform following Relational model into ER Mode id Author name dob AuthorContact id contact Manager writes authorid bookid Book bookid title edition libid memberld location opening Time...
-
The 13 C NMR spectrum of 2-ethylbutanal consists of the following absorptions: 11.5, 21.7, 55.2, and 204.7. Draw the structure of this aldehyde, label each chemically nonequivalent set of...
-
Outline two Wittig alkene syntheses of 2-methyl-1-hexene. Is one synthesis preferred over the other? Why?
-
A silicon diode, whose current-voltage characteristics are given in Fig. 29-30, is connected in series with a battery and a 960-Ω resistor. What battery voltage is needed to produce a...
-
Can you have an effective introduction of a Grant Proposal without using all of the six moves used in writing an introduction of a Grant Proposal? Please explain your response
-
Explain in detail Tim Hortons Branding, Marketing, Market segment, Market share, System Process and Management style. (No word limit) 2. Why did this technology fail? Explain in detail with...
-
Explain how Alba CEO Tim Murray was able to assume an authoritative approach to dealing with the Alba crisis, while a Bahraini executive might not have been as directive and authoritative in dealing...
-
[8] 5. Factor the following simple trinomials. a) p + 11p +30 X
-
Calculate the percent by mass of nitrogen in Cr(NO3)3. Enter a number without the %' symbol
-
Why do product-costing systems based on a single. Volume based cost driver tend to over cost high-volume products? What undesirable strategic effects can such distortion of product costs have?
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
Compound A has the molecular formula C6H12O3 and shows a strong IR absorption peak at 1710 cm-1. When treated with iodine in aqueous sodium hydroxide, A gives a yellow precipitate. When A is treated...
-
The following is an example of a reaction sequence developed by Derin C. D'Amico and Michael E. Jung (UCLA) that results in enantiospecific formation of two new chirality centers and a carbon-carbon...
-
Additional evidence for the halogenation mechanisms that we just presented comes from the following facts: (a) Optically active 2-methyl-1-phenylbutan-1-one undergoes acid-catalyzed racemization at a...
-
Yarianny wants to withdraw $25,000 annually starting today for the next 20 years and will increase the withdrawals by 3.5% each year. If the annuity can earn 6% compounded semi- annually, how much...
-
$1,000 beginning of the quarter deposits will be made into an account for 13 years and 9 months ( 13.75 years ). What was the interest rate compounded semi-annually associated with the account if...
-
December 31 Number Account Title 101 Cash 126 Supplies 128 Prepaid insurance 167 Equipment Debit $ 6,500 34,200 7,500 45,000 Credit 168 Accumulated depreciation-Equipment $ 22,500 173 Building...

Study smarter with the SolutionInn App


