Explain how you would distinguish each of the following isomeric compounds from the others using NMR spectroscopy.
Question:
Explain how you would distinguish each of the following isomeric compounds from the others using NMR spectroscopy. Be explicit.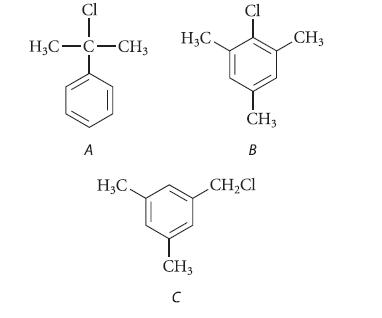
Transcribed Image Text:
Cl T H3C-C-CH3 A H₂C. H₂C. CH3 C Cl CH3 B CH₂Cl CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Only compound A should have a proton NMR spectrum containi...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How could 1H NMR distinguish between the compounds in each of the following pairs? a. CH3CH2CH2OCH3 and CH3CH2OCH2CH3 b. BrCH2CH2CH2Br and BrCH2CH2CH2NO2 c. d. e. f. g. h. i. CH3 CH, CHa CH3CH CHC...
-
How would you distinguish among the compounds within each of the following sets using their NMR spectra? Explain carefully and explicitly what features of the NMR spectrum you would use. (a)...
-
How would you differentiate between the compounds in each of the following pairs? (a) p-ethylbenzoic acid and ethyl benzoate by IR spectroscopy (b) N-methylpropanamide and N-ethyl acetamide by proton...
-
Suppose that the Medicare rate of hospital reimbursement is reduced. Explain why the costs may not be shifted to other patients in the short run.
-
Calculation of tax rafts A multinational computer equipment manufacturer reported the following amounts for t recent years 4in millions of U.S. dollars). The firm applies U.S. GAAP. a. Compute the...
-
Books-For-All is a well-established chain of 20 bookstores in western Ohio. In recent years, the company has grown rapidly, adding five new stores in regional malls. The manger of each store selects...
-
You build a \(2.0-\mathrm{kg}\) mound of mashed potatoes on a kitchen scale (which is a spring scale). You then use a large spoon to press on the top of the mound at a constant force of \(3.0...
-
Presented below are two independent transactions. Both transactions have commercial substance. 1. Global Co. exchanged old trucks (cost $64,000 less $22,000 accumulated depreciation) plus cash of...
-
Provide examples of organizations making the following levels of decisions: strategic decisions, tactical decisions, and operational decisions. What was the result of these decisions? Detail the...
-
(a) Arrange the three isomeric dichlorobenzenes in order of increasing dipole moment (smallest first). (b) Assuming that the dipole moment is the principal factor governing their relative boiling...
-
Which of the following compounds cannot contain a benzene ring? How do you know? C10H16 A C8H6Cl B C5H4 C C10H15N D
-
Indicate the metric unit of measurement that you would use to express the following. The length of an adult whale.
-
A potentially negative uncertainty is known as a(n) ________________, while a positive uncertainty is known as a(n) ________________.
-
In a fixed-price contract, who assumes the greatest level of risk?
-
Which contract type puts the most risk on the seller? a. time and material b. cost reimbursable c. firm-fixed-price d. fixed-price-economic-price-adjustment
-
Who is best known for creating the Quality Trilogy?
-
List and describe the four different categories of project success measures.
-
Suppose a large insurance company wants to estimate the difference between the average amount of term life insurance purchased per family and the average amount of whole life insurance purchased per...
-
What is removed during each of the three stages of wastewater treatment: primary, secondary, and tertiary? During which state would you expect items to be recovered that were accidentally flushed,...
-
Using the Huckel 4n + 2 rule, determine whether each of the following compounds is likely to be aromatic. Explain how you arrived at the -electron count in each case. (a) (b)
-
The following compound is not aromatic even though it has 4n + 2 electrons in a continuous cyclic array. Explain why this compound is not aromatic.
-
Rank the isomers within each set in order of increasing heat of formation (lowest first). CH-CH,
-
Besides requisite financial ability, what other criteria must the Sponsor satisfy?
-
Write the rate law for each of the following elementary reaction equations. State the overall order of the reaction and the order with respect to each reactant. Classify the reaction as unimolecular,...
-
You are the chief of a large city agency. Your department is working with the Federal government through the Secure Communities program. Your city has been declared a "sanctuary" city and has...

Study smarter with the SolutionInn App


