Give an explanation for each of the following facts. (a) The barrier to internal rotation about the
Question:
Give an explanation for each of the following facts.
(a) The barrier to internal rotation about the N-phenyl bond in N-methyl-p-nitroaniline is considerably higher (42–46 kJ mol–1, or 10–11 kcal mol–1) than that in N-methylaniline itself (about 25 kJ mol–1, or 6 kcal mol–1).
(b) Cis- and trans-1,3-dimethylpyrrolidine rapidly interconvert.
(c) CH3NH—CH2—NHCH3 is unstable in aqueous solution.
(d) The following compound exists as the enamine isomer shown rather than as an imine: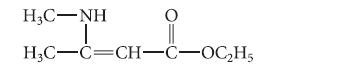
(e) Diazotization of 2,4-cyclopentadien-1-amine gives a diazonium salt, which, unlike most aliphatic diazonium ions, is relatively stable and does not decompose to a carbocation.
Transcribed Image Text:
HỌC…NH | Hi-OGH₂ H3C-C=CH-C-OC₂H5
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a The barrier to internal rotation about the Nphenyl bond in Nmethylpnitroaniline is greater because ...View the full answer

Answered By

Hassan Imtiaz
The following are details of my Professional Experience. Responsibilities Eight years of demanding teaching experience in the field of finance and business studies at Master’s Level. Completion of the given tasks within given time with quality and efficiency. Marketing professional with practical experience in and solid understanding of a diverse range of management applications, including market analysis, sales and marketing, team building and quality assurance. I have excellent skills to approach deal and sustain corporate clients / customers by demonstrating not only extraordinary communication and interpersonal skills but also high caliber presentation, negotiation and closing skills. Manage and follow up the day-to-day activities. Manage and co-ordinate the inventories. Fulfillment of all the tasks assigned.
The following are details of my Areas of Effectiveness. Finance 1. Corporate Finance 2. Advanced Corporate Finance 3. Management of Financial Institutions 4. International Financial Management 5. Investments 6. Fixed Income 7. Real Estate Investment 8. Entrepreneurial Finance 9. Derivatives 10. Alternative Investments 11. Portfolio Management 12. Financial Statement Analysis And Reporting (US GAAP & IFRS) 13. International Financial Markets 14. Public Finance 15. Personal finance 16. Real estate 17. Financial Planning Quantitative Analysis 1. Time Value Of Money 2. Statistics 3. Probability Distribution 4. Business Statistics 5. Statistical Theory and Methods Economics 1. Principles of Economics 2. Economic Theory 3. Microeconomic Principles 4. Macroeconomic Principles 5. International Monetary Economics 6. Money and Banking 7. Financial Economics 8. Population Economics 9. Behavioral Economics International Business 1. Ethics 2. Business Ethics 3. An introduction to business studies 4. Organization & Management 5. Legal Environment of Business 6. Information Systems in Organizations 7. Operations Management 8. Global Business Policies 9. Industrial Organization 10. Business Strategy 11. Information Management and Technology 12. Company Structure and Organizational Management Accounting & Auditing 1. Financial Accounting 2. Managerial Accounting 3. Accounting for strategy implementation 4. Financial accounting 5. Introduction to bookkeeping and accounting Marketing 1. Marketing Management 2. Professional Development Strategies 3. Business Communications 4. Business planning 5. Commerce & Technology Human resource management 1. General Management 2. Conflict management 3. Leadership 4. Organizational Leadership 5. Supply Chain Management 6. Law 7. Corporate Strategy Creative Writing 1. Analytical Reading & Writing Other Expertise 1. Risk Management 2. Entrepreneurship 3. Management science 4. Organizational behavior 5. Project management 6. Financial Analysis, Research & Companies Valuation 7. And any kind of Excel Queries
4.80+
150+ Reviews
230+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give an explanation for each of the following facts. (a) Cis- and trans-1.3-dimethylpyrrolidine rapidly interconvert. (b) The following compound exists as the enamine isomer shown rather than as an...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
How does a project manager calculate start and finish times?
-
This exercise is an extension of Exercise 2-3A. Assume Alice Stern completed the following additional transactions during February. Show the effect of each transaction on the basic elements of the...
-
What circumstances might motivate an audit firm to hire an investigative agency to research a potential client?
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the internal rate of return of this...
-
Jeter Co. uses a perpetual inventory system and both an accounts receivable and an accounts payable subsidiary ledger. Balances related to both the general ledger and the subsidiary ledger for Jeter...
-
2. Please using k-means clustering method to cluster these data points in to 2 clusters. Please provide step by step operations. (10 points) Sample: Feature 1 Feature Step1: 1 1234567 2.5 1.5 2 2.0...
-
Imagine that you have been given a sample of racemic 2-phenylbutanoic acid. Outline steps that would allow you to obtain pure samples of each of the following compounds from this starting material...
-
Outline a sequence of reactions that would bring about the conversion of aniline into each of the following compounds. (a) Benzylamine (b) Benzyl alcohol (c) 2-phenylethanamine (d)...
-
The management of a health insurance company wants to know the percentage of its policyholders who have tried alternative treatments (such as acupuncture, herbal therapy, etc.). A random sample of 24...
-
Company A owns 100% of Company T. Provided below are the sales of Company T and the consolidated group for the past 3 years. Year 1 Year 2 Year 3 Company T Sales 500 800 1,200 Consolidated Sales 800...
-
An FCC iron-carbon alloy has an initial uniform carbon concentration of 0.25 wt% C. The alloy is carburized at 950 C and in an atmosphere that gives a constant surface carbon concentration constant...
-
John Whitten is one of the physicians on staff at Metropolis Health System. His practice is six years old. He has set up an office savings account to accumulate the funds to replace equipment in his...
-
You are using the Adjusted Present Value approach to value the Greenside Co., and you have determined that the value of the unlevered firm is $22,105,137. You calculate the projected interest tax...
-
1. A bag contains 8 red marbles, 8 white marbles, and 8 blue marbles. What is the minimum number of marbles you have to choose randomly from the bag to ensure that we get 3 marbles of same color? 2....
-
Consider the Amazon.com website. The management of the company decided to extend its Webbased system to include products other than books (e.g., wine, specialty gifts). How would you have assessed...
-
Find the equations of the ellipses satisfying the given conditions. The center of each is at the origin. Passes through (2, 2) and (1, 4)
-
Albuterol, shown here, is a commonly prescribed asthma medication. For either enantiomer of albuterol, draw a three-dimensional formula using dashes and wedges for bonds that are not in the plane of...
-
(a) Write the structure of 2 ,2-dichlorobicyclo [2.2.1] heptane. (b) How many chirality centers does it contain? (c) How many stereoisomers are predicted by the 2n rule? (d) Only one pair of...
-
Write appropriate structural formulas for (a) A cyclic molecule that is a constitutional isomer of cyclohexane, (b) Molecules with the formula C6H12 that contain one ring and that are enantiomers of...
-
Who are the staff members from whom you would collect useful information and requirements of store keeper's job?
-
Y5 Question 2. Formulate and explain the duality result between indirect utility and expenditure of a consumer with rational, continuous and strictly convex preferences. [12.5 marks]
-
Blum's algorithm is invoked with k=28 on the following numbers, which means we want to find the 28th smallest element. What is k after one level of recursion? 004 032 140 124 056 041 161 116 128 148...

Study smarter with the SolutionInn App


