Give the principal organic product(s) expected when ethyl benzoate or the other compound indicated reacts with each
Question:
Give the principal organic product(s) expected when ethyl benzoate or the other compound indicated reacts with each of the following reagents.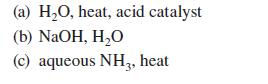
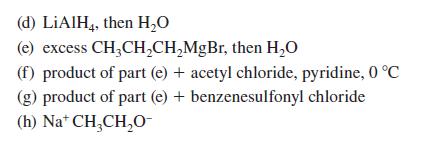
Transcribed Image Text:
(a) H₂O, heat, acid catalyst (b) NaOH, H₂O (c) aqueous NH3, heat
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
PhCOH CH3CHOH e OH PhCCHCHCH3 CHCHCH3 b f PhCO Nat CH3CHOH OCCH3 ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the principal organic product(s) expected when 2- methylthiophene or other compound indicated reacts with each of the following reagents. (a) HNO3 (b) Dilute aqueous NaOH (c) Product of part (a)...
-
Give the principal organic product(s) expected when 2- methylpyridine or other compound indicated reacts with each of the following reagents. (a) Diliute aqueous NaOH (b) HNO3, H2SO4, heat; then -OH...
-
Give the principal organic product(s) expected when p-chloroaniline or other compound indicated reacts with each of the following reagents. (a) Dilute HBr (b) CH 3 CH 2 MgBr in ether (c) NaNO 2 ,...
-
Limits from graph In this problem we evaluate limits from the graph. Consider the graph of f given in [Figure 1]. Evaluate each of the following limits (or explain why if the limit does not exist)....
-
Consider the following transactions that occurred in February 2012 for Gems, Inc. Feb 3 Purchased inventory on terms 1/5, n/eom, $2,000. 4 Purchased inventory for cash of $1,600. 6 Returned $600 of...
-
Calculate the following: a. Calculate the annual cash flows from a $ 2.5 million, 20-year fixed-payment annuity earning a guaranteed return of 7 percent per year if payments are to begin at the end...
-
Refer to the information in Problem 21-1B. Tohono Companys actual income statement for 2017 follows. Required 1. Prepare a flexible budget performance report for 2017. Analysis Component 2. Analyze...
-
In analyzing legal expense for the Boastman Bottle Company, Mary Little, CPA, observes that the company has paid legal fees to three different law firms during the current year. In accordance with...
-
17. In many cases, a worker's hourly wage depends on the number of hours worked. If a certain number of hours is reached, the pay rate beyond that point increases. When working beyond a certain...
-
Complete the diagram shown in Fig P21.35 by filling in all missing reagents or intermediates. benzene A B HNNH OH, heat Figure P21.35 E NaBH C concd. HBr light Br or N-bromosuccinimide, D 1) Mg,...
-
Which polymer should be more resistant to strong base: nylon-6,6 or the polyester in Eq. 21.73? Explain. ndio HOCHCHOH + HOC- ethylene glycol -COH terephthalic acid -HO heat -OCHCH0-C- poly(ethylene...
-
The manager of a wholesale buyers' club has been told by the home office that 40% of all customers favor a new policy that gives nearly immediate checkout (by moving to the front of the line) for...
-
Consider the following column: 0.5Q EI, L 4 B EI, L A The column has a length 2L and is clamped in point A and simply supported in point B. The bending stiffness is El. The column is loaded by a...
-
W hat charge exists that on a test charge that experience a force of 1 . 4 x 1 0 - 8 N at a point where the electric field intensity strength is 5 . 0 x 1 0 - 4 N / C .
-
Salalah Steel SAOG was about to decide whether to launch a new product In year 2002 sale price per unit is OMR 10 and is expected to increase by 10% every year due to inflation. In year 2002 expenses...
-
The following table shows the option prices of HKMU Inc. common stock (HKMU) on 5 November 2022. The closing price of HKMU on that day was USD208.50 and all stock options will expire on 16 December...
-
Consider 6-bits Successive Approximation. If Vref=4.5V and Vin 2.35V, [3 Maks] then use the iterative approach and find the binary equivalent of the input. Show your work. Direct answer will yield...
-
Houston-based Advanced Electronics manufactures audio speakers for desktop computers. The following data relate to the period just ended when the company produced and sold 42,000 speaker sets: Sales...
-
Prairie Outfitters, Inc., a retailer, accepts paymnent through credit cards. During August, credit card sales amounted to $12,000. The processor charges a 3% fee. Assuming that the credit card...
-
Predict the organic product from each of the following oxidation and reduction reactions. (a) (b) (c) (d) (e) HO HCrO4 OH HO OH 1) LAH (2) aq. H2SO4 NaBH4
-
(a) Although we have described the hydrogenation of an alkene as an addition reaction, organic chemists often refer to it as a "reduction." Use the method described in Section 12.2A for assigning...
-
Predict the major organic product from each of the following reactions. (a) (b) (c) (d) (1) CH MgBr (2) HO (1) MgBr (2) NH,CI, H20 MgBr (1 equiv.) O (1) o (2) H,O (1) CH,CHLi (excess) (2) NH,CI, H2O O
-
(a) (b) (c) Find the work done by the force F(r)=-2yi+3xj-zk in the displacement along the helix r = costi+3sintj+tk in the range of 0 t A solid is bounded by the plane surfaces x = 2, y = 2, y = -x...
-
1 4. Let m be a positive integer. State the definition of lim 1 NX n + 1 + m / m exists). Prove lim = 0; that is, L = 0. n m/m + 1+ m = L (assume the limit
-
3) A 2200 lb automobile is jacked up as shown. Determine the compression force (in lbs) in the jack bar. CM 2.4 ft 2.6 ft 4.7 ft 3.9 ft

Study smarter with the SolutionInn App


