Give the structure of an alkene that would give 2-bromopentane as the major (or sole) product of
Question:
Give the structure of an alkene that would give 2-bromopentane as the major (or sole) product of HBr addition.
(The numbers are for reference in the solution.)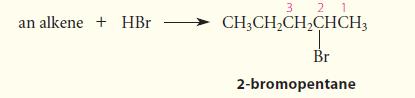
Transcribed Image Text:
an alkene + HBr 3 2 1 CH3CH₂CH₂CHCH3 Br 2-bromopentane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The bromine of the product comes from the HBr However there are many hydrogens in the product Which ...View the full answer

Answered By

Hassan Ali
I am an electrical engineer with Master in Management (Engineering). I have been teaching for more than 10years and still helping a a lot of students online and in person. In addition to that, I not only have theoretical experience but also have practical experience by working on different managerial positions in different companies. Now I am running my own company successfully which I launched in 2019. I can provide complete guidance in the following fields. System engineering management, research and lab reports, power transmission, utilisation and distribution, generators and motors, organizational behaviour, essay writing, general management, digital system design, control system, business and leadership.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each of the following cases, provide the structure of an alkene that would give the alcohol as the major (or only) product of hydroboration--oxidation. CH CH CH CH OH CH, CH,
-
Draw the structure of (a) A five-carbon alkene that would give the same product of HBr addition whether peroxides are present or not. (b) A compound with the formula C6H12 that would not undergo...
-
In each case, give the structure of an eight-carbon alkene that would yield each of the following compounds (and no others) after treatment with ozone followed by dimethyl sulfide. (b)...
-
Debug the given C program to read and print N array elements. Find 10 errors in the given Code and correct the errors. (5Marks) NOTE: Copy and PASTE the correct program in the space below. /*Arrays...
-
1. What tort is at issue here? 2. What are the elements of this tort? 3. Why did the trial court dismiss the case? 4. What did the defendant do here?
-
Access the website for United Way of America ( www.unitedway.org ) and then look through the site until you locate the most recent consolidated financial statements and supplemental schedule and the...
-
Which journal entry records obtaining a bank loan of $15,000? DATE ACCOUNTS POST REF. DR. CR. a. Notes Payable 15,000 Accounts Receivable 15,000 b. Notes Payable Cash 15,000 15,000 C. Cash Notes...
-
As financial manager of Britwell Inc., you are investigating a possible acquisition of Salome. You have the basic data given in the following table. You estimate that investors expect a steady growth...
-
In this problem, "USD" refers to the United States dollar and "EUR" refers to the euro. A year ago, the exchange rate was USD1.2/EUR. Today the exchange rate is USD1.4/EUR. The USD has: a.)...
-
Calculate the unsaturation number for each of the formulas in parts (a) and (b) and each of the compounds in parts (c) and (d). (a) C 3 H 4 Cl 4 (b) C 5 H 8 N 2 (c) Methylcyclohexane (d)...
-
Calculate the standard enthalpy difference between the cis and trans isomers of 2-butene. Specify which stereoisomer is more stable. The heats of formation are, for the cis isomer, 27.40 kJ mol 1 ,...
-
Vacation Villas had the following selected transactions. Feb. 1 Signs a $30,000, eight-month, 5%-interest-bearing note payable to CountryBank and receives $30,000 in cash. 8 Sales on account of...
-
Two 20 m UST and one 15 m UST were removed from a site at a gas station. The excavation resulted in in a tank pit of 15m x 7.5 m x 6 m. The in-situ bulk density (before excavation) is 1.8 g/cm. After...
-
Sales (5,800 units) Cost of goods sold: Joplin Company Absorption Costing Income Statement For the Month Ended April 30 Cost of goods manufactured (6,700 units) Inventory, April 30 (900 units) Total...
-
List and discuss three industry-standard methods for evaluating your mutual fund manager's performance. Give one example for each method suggested. List and discuss three potential issues when trying...
-
Why is it important that methods used to review communication strategies to support workplace diversity be effective, be appropriate. Give an example of what can happen if communication methods are...
-
An insurer has liabilities at the end of the year that are normally distributed with mean of $500 million and standard deviation of $50 million. In contrast to question 3, assets are invested in a...
-
The results of a national survey showed that on average, adults sleep 6.9 hours per night. Suppose that the standard deviation is 1.2 hours. a. Use Chebyshevs theorem to calculate the percentage of...
-
On April 29, 2015, Auk Corporation acquires 100% of the outstanding stock of Amazon Corporation (E & P of $750,000) for $1.2 million. Amazon has assets with a fair market value of $1.4 million (basis...
-
Draw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcycloocatne (b) 2-Cyclobutylhexane (c) 1, 2-Dichlorocyclopentane (d) 1, 3-Dibromo-5-methylcyclohexane
-
Name the following cycloalkanes:
-
Name the following substances, including the cis- or trans-prefix: CH2CH3 (b) , (a) H. - - CI
-
What is an example of a resolution in a story? Also What is a draft resolution in MUN?
-
Dr. Deming would often comment that "this [era] will go down as the age of superstitious learning"? In consideration of the prevalence of Me Organizations, how might you explain this comment?
-
Consider the following system of linear equations. x+y-2z = -1 -3x-6y+9z = -9 -x-3y+4z =-7 (a) Use elementary row operations to determine the augmented matrix in reduced row echelon form. (b) How...

Study smarter with the SolutionInn App


