Give the structure of the product expected from the reaction of each of the following compounds with
Question:
Give the structure of the product expected from the reaction of each of the following compounds with benzene in the presence of one equivalent of AlCl3, followed by treatment with water.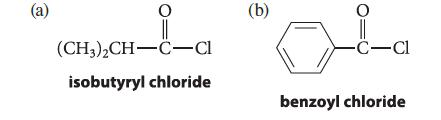
Transcribed Image Text:
(a) 1-1-₁ (CH3)2CH-C-Cl isobutyryl chloride (b) -C-Cl benzoyl chloride
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a CH3CH ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the expected product from the reaction of isopropyl benzene with (a) Hydrogen (3 mol), Pt (b) Sodium and ethanol in liquid ammonia (c) Sodium dichromate, water, sulfuric acid,...
-
Give the structure of the product formed on reaction of ethyl acetoacetate with each of the following: (a) 1-Bromopentane and sodium ethoxide (b) Saponification and decarboxylation of the product in...
-
Give the structure of the product formed on reaction of ethyl acetoacetate with each of the following: (a) 1-Bromopentane and sodium ethoxide (b) Saponification and decarboxylation of the product in...
-
A business had always made an allowance for doubtful debts at the rate of 2 per cent of accounts receivable. On 1 January 2011 the amount for this, brought forward from the previous year, was 300....
-
Accumulated other comprehensive income relations Selected information based on the comparative balance sheets for Solaronx Company (Solaronx), a U.S. defense manufacturer, appears in the following...
-
Glass Corporation began operations on September 1, 2011. The corporations charter authorized 150,000 shares of $8 par value common stock . Glass Corporation engaged in the following transaction...
-
A \(1.2-\mathrm{kg}\) ball dropped from a height of \(3.0 \mathrm{~m}\) onto a steel plate rigidly attached to the ground bounces back up to a height of \(2.5 \mathrm{~m}\). (a) What is the impulse...
-
The City of New York, New York, reported the following liabilities, among others, at June 30 of the years indicated. The amounts in the table are stated in thousands of dollars. None of these...
-
Company X has been experiencing high employee turnover rates in recent years. As a newly hired HR manager, what steps would you take to address this issue and improve employee retention? Identify and...
-
The following compound reacts with AlCl 3 followed by water to give a ketone A with the formula C 10 H 10 O. Give the structure of A and draw a curved-arrow mechanism for its formation. HC- O...
-
A second sulfonation mechanism involves protonated sulfur trioxide as the electrophile. Show the protonation of SO 3 , and draw a curved-arrow mechanism for the reaction of protonated SO 3 with...
-
Calculating Flotation Costs the Educated Horses Corporation needs to raise $40 million to finance its expansion into new markets. The company will sell new shares of equity via a general cash...
-
How is a project transition plan similar to an instruction manual?
-
Improve a work process using either the DMAIC or the PDCA model to guide your actions. What project quality tools did you use, and why did you select each?
-
_______________ is a method for transforming contractual arrangements into a cohesive, collaborative project team with a single set of goals and established procedures for resolving disputes.
-
In your opinion, under what conditions should the sponsor approve a project change, and when is it okay for the project manager to authorize a change? Give an example of each.
-
What is the first step in capturing lessons learned at the end of a project?
-
Test the following hypotheses by using the given sample information and Assume the populations are normally distributed. H0: 21 = 22Ha: 21 22 n1 = 5, n2 = 19, s1 = 4.68, s2 = 2.78
-
What are bounds and what do companies do with them?
-
A method for determining the structures of disubstituted benzene derivatives was proposed in 1874 by Wilhelm Korner of the University of Milan. Kdrner had in hand three dibromobenzenes, A, B, and C,...
-
Give the structures of the principal organic product(s) expected in each of the reactions given in Fig. P16.59 on p. 786, and explain your reasoning. Fig. P16.59 (a) (b) (c) . ACI benzene (large...
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
336 of the Internal Revenue Code, at what value must a liquidating corporation generally be taxed based on the distribution of property when the property has been sold to shareholders?
-
What is the last opportunity for tax shareholders to on any earnings and profits that the corporation has accumulated? Explore the terms acquisition, liquidation and redemption.
-
A bond maturing on Dec 31, 2026. is sold on May 1, 2021. Last coupon date was Dec 31, 2020. Next Coupon date is June 30, 2021. It pays 7% coupon (paid semi-annually). Par value is $1000. Yield is 5%....

Study smarter with the SolutionInn App


