Give the structure of the product in the reaction of each of the following esters with isotopically
Question:
Give the structure of the product in the reaction of each of the following esters with isotopically labeled sodium hydroxide, Na+ 18OH– and explain your reasoning.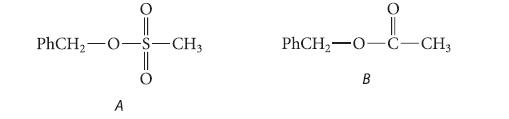
Transcribed Image Text:
PhCH₂-O- A -CH3 PhCH₂-0-C-CH3 B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Because sulfonate esters behave like alkyl halides ...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of the expected organic product in the reaction of 3-phenylpropanal with each of the following: (a) Chlorine in acetic acid (b) Sodium hydroxide in ethanol, 10C (c) Sodium...
-
Give the structure of the expected organic product in the reaction of 3-phenylpropanal with each of the following: (a) Chlorine in acetic acid (b) Sodium hydroxide in ethanol, 10C (c) Sodium...
-
Give the structure of the product formed on reaction of ethyl acetoacetate with each of the following: (a) 1-Bromopentane and sodium ethoxide (b) Saponification and decarboxylation of the product in...
-
1. What options does Personal Trainer have for developing a new system? What are some specific issues and options that Susan should consider in making a decision? 2. Susan has been asked to prepare a...
-
Thelmas Amusements completed the following transactions during November 2012: Nov 1 Purchased supplies for cash, $700. 4 Purchased inventory on credit terms of 3/10, n/eom, $9,600. 8 Returned half...
-
What factors contributed to the significant decrease in profits for securities firms in the early 2000s and the resurgence in profits in the middle of the first decade of the 2000s?
-
By using six factor formula for \(k\), derive the Eqs. (7.93), (7.94) of Section 7.7.1. dkoo dp= k MB dM dB 8 + (7.93) 1+M B M B2
-
The trial balance of Valdez Fashion Center contained the following accounts at November 30, the end of the companys fiscal year. Adjustment data: 1. Supplies on hand totaled $2,000. 2. Depreciation...
-
1 = We know that all the statements [4] Consider the system (0.3)-(0.4) in the particular case f(r) = proven in Problem [3] apply to this case, so for example, we know that c2 7 = - 1 12 (0.6) de and...
-
(a) Draw the structure of the product that is formed when the following compound is heated with one equivalent of sodium methoxide in methanol. Explain your reasoning. (b) What would the product be...
-
Using an acid chloride synthesis as a first step, outline a conversion of hexanoic acid into each of the following compounds. (a) Ethyl hexanoate (b) N-methylhexanamide
-
There are five sales associates at Mid-Motors Ford. The five associates and the number of cars they sold last week are: Sales Associate Cars Sold Peter Hankish....... 8 Connie Stallter ....... 6 Juan...
-
What are process steps during period end closing activities?Explain.
-
A sled is sitting at the top of a frictionless inclined plane, which is 5.2 meters long with an angle of 30.0 slope above the horizontal. What will be the velocity of the sled when it reaches the...
-
BigBusiness Inc. is looking solely at the amount of money that they need to bring in next year. What financial planning requirements is BigBusiness focusing on?
-
What is the implication of the absolute version of purchasing power of parity? Explain
-
Molly received a $2.00 off coupon for a $10.00 product. After redeeming the coupon, she paid $8.00 for the product and saved $2.00. Millie received a $2.00 rebate for a $10.00 product. She paid...
-
A contribution income statement for the Nantucket Inn is shown below. (Ignore income taxes.) Revenue ..........................$500,000 Less: Variable expenses .................... 300,000...
-
Carlton Stokes owns and operates a car-detailing business named SuperShine & Detailing. For $150, Carltons business will hand wash and wax customers cars, vacuum the interior, and thoroughly clean...
-
Write a retrosynthetic analysis and then synthetic reactions that could be used to prepare racemic Meparfynol, a mild hypnotic (sleep-inducing compound), starting with compounds of four carbon atoms...
-
Write a retrosynthetic analysis and synthesis for the following transformation. OH Br HO HO
-
Synthesize the following compound using cyclopentane and ethyne (acetylene) as the sole source of carbon atoms.
-
Problem 6 (6 points) Consider the sequence of continuous functions {fn} on [0, 1] given by fn(2): = {19 nx 0x <1/n 1/n x 1 Show that {f} has no subsequence which is convergent in uniform norm. (Hint:...
-
Problem 1. [10pts] Consider the weights vector r = , 12 = , 13 = to define (, )1 11 the inner product with weight in R (as seen in class). Let --(0)-0 3 2 y = 4 1. (2pts) Show that x and y are...
-
Break-even analysis requires these three components to calculate it. Responses gross margin percent, shrinkage and rate of sale. gross margin percent, shrinkage and rate of sale. gross margin...

Study smarter with the SolutionInn App


