How many moles of permanganate are required to oxidize one mole of toluene to benzoic acid? (Use
Question:
How many moles of permanganate are required to oxidize one mole of toluene to benzoic acid? (Use H2O and protons to balance the equation.)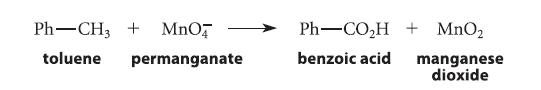
Transcribed Image Text:
Ph-CH3 + MnO toluene permanganate Ph-CO₂H + MnO₂ benzoic acid manganese dioxide
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The glib way to work this problem is to balance the changes in oxidation states ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many moles of dichromate are required to oxidize one mole of ethanol to acetaldehyde? ethanol dichromate acetaldehyde
-
Tranquil Teas makes bottled iced teas and uses a process cost system. Each day, Tranquil produces 250 bottles of tea; each period, Tranquil produces 23,000 bottles; each year, Tranquil produces...
-
Use the solution of Problem 19-12 to calculate how many moles of photons of UV light ( = 220 nm) would be required to drive the four-electron oxidation of H2O by NADP+ under standard conditions to...
-
Selected answer is incorrect During substantive procedures, performing analytical procedures satisfies which primary audit objective: Cutoff Accuracy Existence Completeness 2 answers
-
Refer to the data in PE 6-18. Assume that during the following year, 20 customers bring in 80 tires for warranty repairs. The labor and supplies associated with these repairs were $900 and $350,...
-
List four general cases giving rise to temporary differences between financial reporting and tax reporting. Discuss.
-
What is the main difference between core and supplemental ecological indicators? Provide an example of each.Would it be useful to have all production site managers to report to top management on a...
-
The unadjusted trial balance of PS Music as of July 31, 2016, along with the adjustment data for the two months ended July 31, 2016, are shown in Chapter 3. Based upon the adjustment data, the...
-
Assume that your company is considering a project that has an up-front cost of $1,500,000 at Year O. The project's subsequent cash flows are uncertain: there is a 75 percent chance that the cash...
-
Write a curved-arrow mechanism for the following oxidation of 2-heptanol, which proceeds in 82% yield. OH 2-heptanol + Ph3C+ BFT a relatively stable carbocation 2-heptanone + HBF4 + Ph3CH
-
Contrast the products expected when 2-methyl-3-pentanol is treated with (a) HBr/H 2 SO 4 or (b) Ph 3 PBr 2 . Explain.
-
What two roles are played by accountants with respect to the information system?
-
Explore the website of the UN Global Compact initiative, at https://www.unglobalcompact.org/. Click on the heading Engage Locally, and explore some of the companies in your part of the world that...
-
A horizontal metal strip \(1.0 \mathrm{~mm}\) thick and \(20 \mathrm{~mm}\) wide carries a 20-A current along its length, and both the length and the width are perpendicular to a uniform magnetic...
-
A particle that has mass \(m\) and charge \(q\) enters a uniform magnetic field that has magnitude \(B\) and is directed along the \(x\) axis. The initial velocity of the particle is in the \(x y\)...
-
An electron carrying a charge \(-e=-1.60 \times 10^{-19} \mathrm{C}\) moves in a straight line at a speed \(v=3.0 \times 10^{7} \mathrm{~m} / \mathrm{s}\). What are the magnitude and direction of the...
-
The isotopes magnesium-24 (mass \(3.983 \times 10^{-26} \mathrm{~kg}\) ) and magnesium-26 (mass \(4.315 \times 10^{-26} \mathrm{~kg}\) ) are to be separated using a mass spectrometer in which the...
-
Each of the following is a cost incurred by Fairgood & Hernandez, a small CPA firm. REQUIRED Identify whether each of the following costs most likely relates to an (1) organization-sustaining...
-
Find the cross product a x b and verify that it is orthogonal to both a and b. a = (t, 1, 1/t), b = (t 2 , t 2 , 1)
-
What general kind of reaction does NAD+ carry out?
-
What general kind of reaction does FAD carry out?
-
Why arent the glycolysis and gluconeogenesis pathways the exact reverse of one another?
-
Raymond Supply, anational hardware chain, is considering purchasing a smaller chain,Strauss & Glazer Parts (SGP). Raymond\'s analysts project thatthe merger will result in the following free cash...
-
Vincent has also been working with Jennifer to sell her rental property and to buy a new house in a nearby suburb. Vincent's real estate agency manages the property on behalf of Jennifer and collects...
-
How does emotion regulation influence individuals' responses to conflict, and what techniques can be employed to cultivate emotional intelligence and self-awareness to facilitate constructive...

Study smarter with the SolutionInn App


