Identify the benzylic carbons in each of the following structures. (a) H3C CH3 (b) HC
Question:
Identify the benzylic carbons in each of the following structures.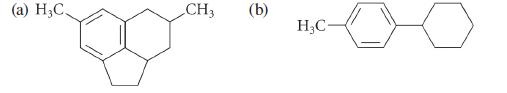
Transcribed Image Text:
(a) H3C CH3 (b) H₂C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The benzylic ...View the full answer

Answered By

FREDRICK MUSYOKI
Professional Qualities:
Solution-oriented.
Self-motivated.
Excellent problem-solving and critical thinking skills.
Good organization, time management and prioritization.
Efficient troubleshooting abilities.
Tutoring Qualities:
I appreciate students as individuals.
I am used to tailoring resources for individual needs.
I can integrate IT into student's lessons.
I am good at explaining concepts.
I am able to help students progress.
I have a wide curriculum knowledge.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the benzylic carbons in the following structures. H,C-
-
Benzylic radicals, due to the adjacent benzene ring, have even greater possibility for de-localization than allylic radicals. Draw contributing resonance structures that show this delocalization for...
-
Identify the compound with molecular formula C6H10O that is responsible for the following DEPT 13C NMR spectrum. CH3 carbons CH2 carbons CH carbons all protonated carbons 200 180 160 140 120 100 80...
-
During extension, why do we sometimes get joints (and veins) and other times shear fractures and faults?
-
Effect of rent transactions on statement of cash flows A firm reported a balance in its Pre paid Rent (Advances to Landlord) account of $1,200 on January 1, 2009, for use of the building for the...
-
The trial balance for Marek Services at the end of July 2011 appeared at the Marek's accountant compared the amounts in the trial balance with the ledger, recomputed the account balances, and...
-
The molecules in the rods and cones in the eye are tuned to absorb photons of particular energies. The retinal molecule, like many molecules, is a long chain. Electrons can freely move along one...
-
Prepare a list of activities for a systems development team for an online travel agent that is setting up a Web site for customers. Now suppose you are running out of time. Describe some of your...
-
In the article " The Framework, Concepts, and Methods of the Competency Outcomes and Performance Assessment (COPA) Model" (new tab)opens a new window , the authors emphasize the importance of having...
-
Predict the order of relative reactivities of the compounds within each series in S N 1 solvolysis reactions, and explain your answers carefully. (a) (b) Cl T -CH- CH3 (1) CH3 of CH3 (1) -C-Cl CHO...
-
What products are expected in the reaction of H 2 C = CHCH 2 CH 2 CH 2 CH 3 (1-hexene) with NBS in CCl 4 in the presence of peroxides? Explain your answer.
-
Annual starting salaries for college graduates with degrees in business administration are generally expected to be between $45,000 and $60,000. Assume that a 95% confidence interval estimate of the...
-
Indicate whether the following expenditures are trade or business deductions (T), production of income deductions (PI), personal deductions (P), or are not deductible (X). Also indicate if the...
-
Mr. Leff received a $6,200 grant from a local university for the fall of 2019, Mr. Leff was a candidate for a degree, and was required to be a research assistant, for which services he received...
-
Indicate whether the following expenditures are deductible for AGI, from AGI or not deductible. a. Medical expenses of individual taxpayer b. Safe deposit box rental for business c. Interest expense...
-
David and Renee Kimberly, ages 75 and 65, respectively, have the following sources of income: Their itemized deductions total $11,000. Compute their taxable income. Private pension receipts Social...
-
In 2019, Windsor Knott, an employee of the Victoria Tie Company, was seriously injured in the factory stockroom. He was hospitalized for 30 days and lost partial use of his left hand. During his...
-
Why do employees "blow the whistle"on other employees for unethical or illegal behavior? One study conducted by the AICPA reported the likelihood that employees would blow the whistle on another...
-
An investor sells a European call on a share for $4. The stock price is $47 and the strike price is $50. Under what circumstances does the investor make a profit? Under what circumstances will the...
-
A compound A has a strong, broad IR absorption at 3200-3500 cm-1 and the proton NMR spectrum shown in Fig. P13.48a. Treatment of compound A with H2SO4 gives compound B, which has the NMR spectrum...
-
What is the chemical-shift difference in ppm of two resonances separated by 45 Hz at each of the following operating frequencies? (a) 60 MHz (b) 300 MHz
-
(a) How many different sets of proton absorptions (ignoring splitting) should be observed in the proton NMR spectrum of 4-methyl-l-penten-3-ol? (b) How many absorptions should there be in the 13C NMR...
-
How the explain the effect on immigration law, non-citizens and U.S. citizens?
-
In order to buy new equipment, your company took a $150,000 loan to be paid over 15 years with a nominal annual rate of 2.5%. You make monthly payments, at the end of each month. 1. How much interest...
-
How do you estimate the kernel density of a random variable?

Study smarter with the SolutionInn App


