Identify the compound C 4 H 9 NO with the proton NMR spectrum given in Fig. 21.3.
Question:
Identify the compound C4H9NO with the proton NMR spectrum given in Fig. 21.3. This compound has IR absorptions at 3300 and 1650 cm–1.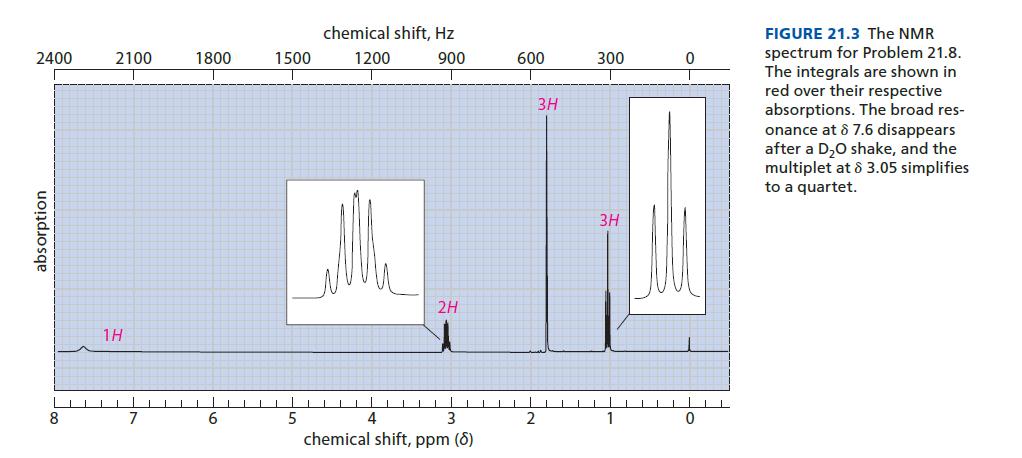
Transcribed Image Text:
2400 absorption 8 2100 18 7 1800 6 1500 5 chemical shift, Hz 1200 900 T 2H 4 3 chemical shift, ppm (8) 600 2 3H 300 3H 1 0 0 FIGURE 21.3 The NMR spectrum for Problem 21.8. The integrals are shown in red over their respective absorptions. The broad res- onance at 8 7.6 disappears after a D₂O shake, and the multiplet at 8 3.05 simplifies to a quartet.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
That the compound is an amide is confirmed by the NH ab...View the full answer

Answered By

Sumit kumar
Education details:
QUATERNARY Pursuing M.Tech.(2017-2019) in Electronics and Communication Engg. (VLSI DESIGN) from
GNIOT Greater Noida
TERTIARY B.Tech. (2012-2016) in Electronics and Communication Engg. from GLBITM Greater Noida
SECONDARY Senior Secondary School Examination (Class XII) in 2012 from R.S.S.Inter College, Noida
ELEMENTARY Secondary School Examination (Class X) in 2010 from New R.J.C. Public School ,Noida
CERTIFICATION
Summer Training in ‘WIRELESS EMBEDDED SYSTEM’ from ‘XIONEE’ for the six weeks.
EMBEDDED SYSTEM Certificate issued by CETPA INFOTECH for one day workshop.
Certificate of Faculty development program on OPTICAL COMMUNICATION and NETWORKS for one week.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the compound with the mass spectrum and proton NMR spectrum shown in Fig. P19.67. This compound has IR absorptions at 1678 cm 1 and 1600 cm 1 . 100 183 185 155 60 40 1157 43 198 200 La fl 0...
-
(a) Propose a structure for an amine A (C 4 H 9 N), which liberates a gas when treated with NaNO 2 and HCl. The 13 C NMR spectrum of A is as follows, with attached protons in parentheses: 14(2), ...
-
Propose structures for compounds that meet the following descriptions: (a) C5H8, with IR absorptions at 3300 and 2150 cm1 (b) C4H80, with a strong IR absorption at 3400 cm1 (c) C4H80, with a strong...
-
Write C++ statements to do the following. a. Declare int variables num1 and num2. b. Prompt the user to input two integers. c. Input the first number in num1 and the second number in num2. d. Output...
-
Review the data given in Exercise 5-21. Requirement 1. Prepare Budgets single-step income statement.
-
Consider the following income statement for WatchoverU Savings Inc. (in millions): a. What is WatchoverUs expected net interest income at year-end? b. What will be the net interest income at year-...
-
An effective way to learn how companies respond to the competing pressures to be globally integrated and locally responsive is to study them in action. Referring back to Exhibit 6.3, search online...
-
Colter Company prepares monthly cash budgets. Relevant data from operating budgets for 2013 are: All sales are on account. Collections are expected to be 50% in the month of sale, 30% in the first...
-
Simplify the given expression and write the answer with only positive exponents. -113 P 3/7 7 3 54x y z 2 w) 3) (w + 2) (3w + w 4 2 (-3x +7x+8)= (x * + 7x *-12x-1) - 5 27-7-28 20-x-22 712-24-8...
-
(a) Assuming that the difference in the relative boiling points of methyl acetate and 2-butanone (see display above) is caused by the difference in their dipole moments, predict which compound has...
-
Draw the structure of an amide that must exist in an E conformation about the carbonylnitrogen bond.
-
Solve graphically using a calculator: 4x 2 5x 3 = 0
-
What must take place to ensure the success of a CRM system? Explain
-
Which entry correctly shows John paying the contractor $1,500 cash for the future expense of replacing a section of the roof?
-
. We want to compute the derivative of the following function. g(x): = e5x2 (tan(x))3 24 a) Use the properties of the logarithmic functions to simplify the formulation of In g(x)|. Answer: In g(x) =...
-
The taxable benefit for an interest-free or low-interest loan is calculated using which rate?
-
What are the implications of institutional voids for doing business in emerging markets? Explain
-
College Pizza delivers pizzas to the dormitories and apartments near a major slate university. The companys annual fixed expenses are $40,000. The sales price of a pizza is $10, and it costs the...
-
B made an issue of 150,000 $1 ordinary shares at a premium of 20% the proceeds of which is received by cheque. What is the correct journal to record this? A. Bank Share capital Share premium B. Bank...
-
Which diene and dienophile would you employ to synthesize the following compounds? (a) (b) CO2Me CO2Me
-
Diels-Alder reactions also take place with triple-bonded (acetylenic) dienophiles. Which diene and which dienophile would you use to prepare the following? CO2Me CO2Me
-
1,3-Butadiene and the dienophile shown below were used by A. Eschenmoser in his synthesis of vitamin B12 with R. B. Woodward. Draw the structure of the enantiomeric Diels-Alder adducts that would...
-
I use spreadsheets daily for both my personal and work life. I use them to calculate child support debt for non-custodial parents who owe a debt to the state, tribal, or non-assist. This helps us...
-
CMS 1500 billing form requirements. Students will audit an electronic claim in EHR Go and identify any missing fields that could cause claims to be rejected
-
Economic value added is used to _________________. Group of answer choices measure the company's profits measure the company's expenses None of the above measure the company's sales

Study smarter with the SolutionInn App


