In each case, give the structure of a compound with the indicated formula that would give the
Question:
In each case, give the structure of a compound with the indicated formula that would give the following diol in a LiAlH4 reduction followed by protonolysis.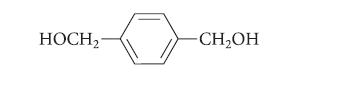
(a) C8H6O3
(b) C8H6O4
Transcribed Image Text:
HOCH2 -CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a The compound must contain three oxygens and two degrees of unsaturation in addition to ...View the full answer

Answered By

Subash Murugaih
I am leading expert in this web site couple of years and My clients are much happy with my works and services.
4.60+
309+ Reviews
539+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the structure of a compound with the indicated formula that would give the following diol in a LiAlH4 reduction followed by protonolysis. C8H6O4 HOCH,- -CH,OH
-
Give the structure of a compound that satisfies each of the following criteria. (a) A compound C3H7ON that liberates ammonia on treatment with hot aqueous KOH (b) A compound that gives equal amounts...
-
Give the structure of a compound that fits each description. (There may be more than one correct answer for each.) (a) A chiral primary amine C4H7 N with no triple bonds (b) A chiral primary amine...
-
List the following classifications of accounts in all of the columns in which they appear on the work sheet, with the exception of the Adjustments columns. (Example: Assets) Assets ...................
-
Matthews Delivery Service, Inc., completed the following transactions during its first month of operations for January 2012: a. Matthews Delivery Service, Inc., began operations by receiving $6,000...
-
Consider the following cumulative relative frequency distribution. Class .......... Cumulative Relative Frequency 150 up to 200 ...... 0.10 200 up to 250 ...... 0.35 250 up to 300 ...... 0.70 300 up...
-
The company is committed to ethical conduct and has no tolerance for fraud and unethical behavior. There are some concerns about abuses in your department. Do you know anything about the concerns I...
-
Polk Company builds custom fishing lures for sporting goods stores. In its first year of operations, 2012, the company incurred the following costs. Variable Cost per Unit Direct materials...
-
A 25-year-old Latin, G1P1, cisfemale presents to the office with a 6-month history of amenorrhea; reports having regular menstrual cycles every month up until about 1 year ago when her menstrual...
-
Give the structures of the products formed when (a) Chloroacetic acid and (b) P-chloro ben zoic acid react with P 2 O 5 .
-
Give the structure of the acid chloride formed in each of the following transformations. (a) Sodium ethanesulfonate + PCl 5 (b) Benzoic acid + SOCl 2 (c) P-toluenesulfonic acid 1 excess...
-
Which statement is correct? A. There is a resultant force on an object moving along a circular path at constant speed away from the centre of the circle causing it to be thrown outwards. B. There is...
-
Talcon Corporation has a 11% unlevered cost of equity. The company forecasts the free cash flows shown below. The cash flows are expected to grow at a constant rate of 4% rate after Year 3. Unlevered...
-
The risk free rate is 2% and the market's risk premium is 6%. If the CAPM holds, what is the beta of a security with a 12% expected return?
-
Train cars are coupled together by being bumped into one another. Suppose two loaded train cars are moving toward one another, the first having a mass of 130,000 kg and a velocity of 0.300 m/s, and...
-
For each of the following transactions, explain why they would or would not be counted in the GDP of the United States. a. Japanese auto producer Honda builds a factory in Indiana. b. You buy a new...
-
A project will require $543,000 for fixed assets, $118,000 for inventory, and $142,000 for accounts receivable. Short - term debt is expected to increase by $65,000. The project has a six-year life....
-
Piscataway Plastics Company manufactures a highly specialized plastic that is used extensively in the automobile industry. The following data have been compiled for the month of June. Conversion...
-
Organizations are increasing their use of personality tests to screen job applicants. What are some of the advantages and disadvantages of this approach? What can managers do to avoid some of the...
-
When 1,3,5-cycloheptatriene reacts with one molar equivalent of bromine at 0 8C, it undergoes 1,6 addition. (a) Write the structure of this product.
-
The cyclopentadienyl cation is apparently antiaromatic. Explain what this means in terms of the p-electron energies of a cyclic and an open-chain compound.
-
In 1967 R. Breslow (of Columbia University) and co-workers showed that adding SbCl5 to a solution of 3-chlorocyclopropene in CH2Cl2 caused the precipitation of a white solid with the composition C3H3...
-
O the approximate values obtained in Problem 1. Problem 3 (20 points). The X-15 hypersonic research airplane set the altitude record for airplanes on August 22, 1963, when test pilot Joseph Walker...
-
Dale, a 40 year old single taxpayer, incurs medical expenses for the year as follows: Medical insurance premiums: $11,000 Doctors and hospital fees: $3,500 Hearing aids : $ 350 General purpose...
-
Question #2 ON 6 BOUBLE ACTING CYLINDER + A JULY PART MOMODEL TYPE 1MTV-3 TOGGAL 2.3MAV-3 AYT. 4,9M6v1 MFC-2 SHUTTLE 7MAT-30AM TANK MAPS ACTUA 9 MAY 4 WAYYA 10,11 PUSH BUTTON A. If push button #11...

Study smarter with the SolutionInn App


