In each of the following sets, specify the one compound that is likely to have completely ionic
Question:
In each of the following sets, specify the one compound that is likely to have completely ionic bonds in its solid state.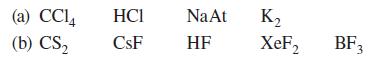
Transcribed Image Text:
(a) CC14 (b) CS₂ HCI CsF Na At HF K₂ XeF₂ BF₁
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a Compound 3 sodium astatide would be most likely to exis...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
In each of the following situations, specify the population. Also, state the two categorical variables that would be measured for each unit in the sample and the two categories for each variable. a....
-
In each of the following independent cases, state whether the bonds were issued at par, a premium, or a discount. Explain your answers. (a) Pop-up Manufacturing sold 1,500 of its $1,000, 8%...
-
The toroid of FIGURE P29.55 is a coil of wire wrapped around a doughnut-shaped ring (a torus). Toroidal magnetic fields are used to confine fusion plasmas. a. From symmetry, what must be the shape of...
-
Selecting Graphics Your Task. Identify the best graphics form to illustrate the following data. a. Figures comparing the costs of cable, DSL, and satellite Internet service in ten major metropolitan...
-
Baker, Inc., purchases office furniture (7-year MACRS property) costing $511,000 and a computer system (5-year MACRS property) costing $511,000 in 2011. Assuming that Baker elects not to claim bonus...
-
What are the three parts of a make rule?
-
For the following independent situations, assume that you are the audit partner on the engagement: 1. In the last 3 months of the current year, Oil Refining Company decided to change direction and go...
-
Write a C program that asks the user to enter the sum and difference of 2 integer numbers. The program will find the 2 numbers and display them as shown below. Input validation: sum plus difference...
-
(a) Construct a hybrid orbital picture for the water molecule using oxygen sp 3 hybrid orbitals. (b) Predict any departures from tetrahedral geometry that you might expect from the presence of two...
-
The bond dissociation energy of H 2 is 435 kJ mol 1 (104 kcal mol 1 ); that is, it takes this amount of energy to dissociate H 2 into its atoms. Estimate the bond dissociation energy of H 2 + and...
-
The risk-averse person will buy health insurance a. Only if the expected health costs equal the insurance premium. b. Only if the expected health costs are greater than the insurance premium. c. Even...
-
Organizational data involved with the fulfillment process are: Material Mater (part number, delivering plants, sales units etc), Customer master data (name, address, account number, payment terms,...
-
"You were there. Why wouldn't the jury think you were involved? Isn't it true, monkey see monkey do?" Critically discuss the above in light of the psychological perspectives relating to ethical...
-
2. What is the difference between vertical and horizontal FDI? Give one example of an industry for each type?
-
State a key difference between the Single Period Model and the other Inventory Models (i.e., EOQ and ROP models). Provide an example of single period products. Provide an example where you see a...
-
Casey deposited $800 at the end of every month into an RRSP for 9 years. The interest rate earned was 4.75% compounded semi-annually for the first 3 years and changed to 5.00% compounded monthly for...
-
User cost is equal to the present value of future profits in the model presented in Figure. Choosing the optimal extraction level Will the optimal quantity to mine in the present year increase or...
-
Refer to the table to answer the following questions. Year Nominal GDP (in billions) Total Federal Spending (in billions) Real GDP (in billions) Real Federal Spending (in billions) 2000 9,817 578...
-
Show all the steps in the mechanism for thisreaction: CH,Br CH2OET ELOH OEt
-
Classify these transformations as oxidations or reductions: a) b) d) CH;CH,OH CH,CH,CH,OH
-
Show a mechanism for thisreaction: OCCH3 obcn, OCCH3 OTS CH,CO,H
-
An undersea research chamber is spherical with an external diameter of 4.10 m . The mass of the chamber, when occupied, is 35200 kg . It is anchored to the sea bottom by a cable. The density of...
-
You would like to pay $22,000 cash for a new car 4 years from today. How much will you need to deposit today in a savings account paying 2% in order to afford the new car in 4 years? Question 6 1 pts...
-
Two rods have the same mass and length, but the mass is distributed differently. One rod has the mass concentrated at the ends and the other has the mass concentrated at the center. The two rods are...

Study smarter with the SolutionInn App


