In the infrared spectrum of nonane in Fig. 12.4, what is the absorbance of the sharp peak
Question:
In the infrared spectrum of nonane in Fig. 12.4, what is the absorbance of the sharp peak at 1380 cm–1?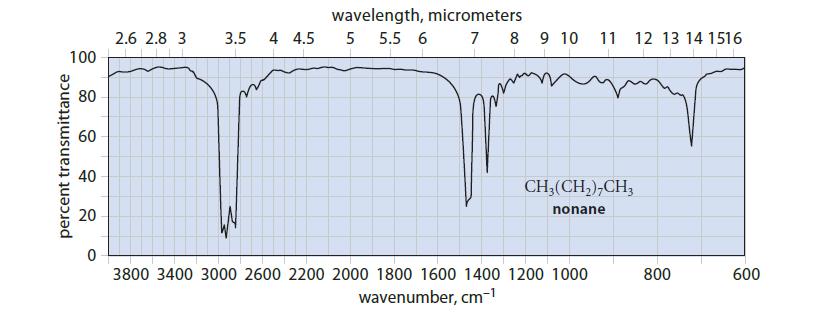
Transcribed Image Text:
percent transmittance 100 80 60 40 20 0 2.6 2.8 3 wavelength, micrometers 3.5 4 4.5 5 5.5 6 7 8 9 10 11 CH3(CH₂)7CH3 nonane 3800 3400 3000 2600 2200 2000 1800 1600 1400 1200 1000 wavenumber, cm-¹ 12 13 14 1516 800 600
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
The peak at 1380 cm has a percen...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
which of the following is not required to be proven to hold a defendant liable for negligent conduct? a. intent to do the harm. b. duty. c. breach of duty. d. physical injury or financial loss. e....
-
Is an eigenvector of If so, find the eigenvalue. 4 -3 1
-
Find the area of each of the shaded regions: yA y = x + 1 1 1
-
Marketing costs like advertising and sales commissions are Multiple choice question. part of costs of goods sold. part of gross profit. selling expenses
-
What is the difference between accounting controls and administrative controls?
-
At the beginning of January, Conway Coffee Bean Shop purchased inventory for $5,000 cash to sell in January and February. The shop sold inventory that cost $2,100 in January and the remainder in...
-
The Tuckers owned an RV that they insured through American Family. On August 26, 2012, their RV was struck by lightning and damaged. The Tuckers reported the damage to American Family. In March 2013,...
-
A company has sales of $1,000,000, variable costs of $250,000, and fixed costs of $600,000. Compute the following: 1. Contribution margin ratio. 2. Break-even sales volume. 3. Margin of safety ratio....
-
What are some practical advantages to using a star schema data model over a relational model in healthcare?
-
Match the IR spectrum in Fig. 12.13 to one of the following three compounds: 2-methyl-1-octene, butyl methyl ether, or 1-pentanol. FIGURE 12.13 The IR spec- trum for Problem 12.13. percent...
-
Each of three alkenes, A, B, and C, has the molecular formula C 5 H 10 , and each undergoes catalytic hydrogenation to yield pentane. Alkene A has IR absorptions at 1642, 990, and 911 cm 1 ; alkene B...
-
The Coca-Cola Company is a global soft-drink beverage company that is a primary and direct competitor with PepsiCo. The data in Chapter 12 Exhibits 12.14-12.16 include the actual amounts for 2012 and...
-
Explain the difference between the nominal rate of interest and the real rate of interest.
-
What federal agency regulates the activities of credit unions?
-
a. What is the primary asset in which savings and loan associations invest? b. Why were banks in a better position than savings and loan associations to weather rising interest rates?
-
A friend has asked you to help him figure out a statement he received from his broker. It seems that, at the start of last year, your friend paid \(\$ 900\) for a bond, and sold it at the end of the...
-
The impedance of a generator, with the rated specifications of \(20 \mathrm{kV}\) and 200 MVA, is \(\mathbf{Z}=j 0.2 p\). \(u\). Determine its reactance in percent if \(21 \mathrm{kV}\) and \(100...
-
Chloe and Emma start a new business, Cement Sidewalks and Accessories (CSA), during the current year. CSA is organized as a partnership. Chloe owns 40% of CSA; Emma owns the remaining 60%. Chloe and...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
Compound A, C 6 H 12 O, has an IR absorption at 1715 cm ?1 and gives compound B, C6H15N, when treated with ammonia and NaBH3CN. The IR and 1 H NMR spectra of B are shown. What are the structures of A...
-
Name the following amines, and identify each as primary, secondary, ortertiary: (b) (a) (c)
-
The following compound contains three nitrogen atoms. Rank them in order of increasing basicity.
-
practice problem accompanying it discuss Differential Cost Analysis. Read through these examples and understand (without concentrating on the math) what these examples are telling us. What is the...
-
Maria is an auditor. She is analyzing a report of monitoring alerts that occurred over time to determine if a pattern of events exists. What is Maria most likely performing?
-
Company K uses LIFO to value its inventory. At the end of the year, how should K measure its inventory under ASC 330?

Study smarter with the SolutionInn App


