In the structure of acetyl-CoA (Fig. 25.1), point out and identify the phosphorus-containing functional groups. HC-C- HC-C-SCOA
Question:
In the structure of acetyl-CoA (Fig. 25.1), point out and identify the phosphorus-containing functional groups.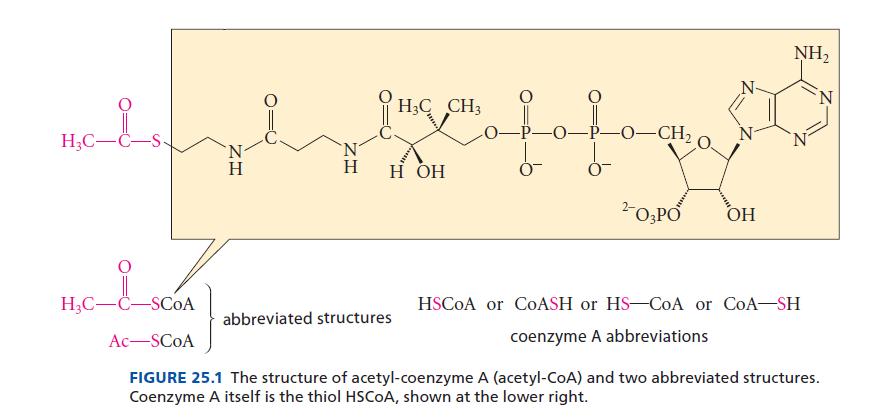
Transcribed Image Text:
H₂C-C- H₂C-C-SCOA Ac-SCOA ZH Η H3C CH3 slave fofagart —O–CH, H H OH 2- O3PO OH abbreviated structures NH₂ HSCOA or COASH or HS-CoA or CoA-SH coenzyme A abbreviations N FIGURE 25.1 The structure of acetyl-coenzyme A (acetyl-CoA) and two abbreviated structures. Coenzyme A itself is the thiol HSCOA, shown at the lower right.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
HgC thioester HC ...View the full answer

Answered By

ANDREW KIPRUTO
Academic Writing Expert
I have over 7 years of research and application experience. I am trained and licensed to provide expertise in IT information, computer sciences related topics and other units like chemistry, Business, law, biology, biochemistry, and genetics. I'm a network and IT admin with +8 years of experience in all kind of environments.
I can help you in the following areas:
Networking
- Ethernet, Wireless Airmax and 802.11, fiber networks on GPON/GEPON and WDM
- Protocols and IP Services: VLANs, LACP, ACLs, VPNs, OSPF, BGP, RADIUS, PPPoE, DNS, Proxies, SNMP
- Vendors: MikroTik, Ubiquiti, Cisco, Juniper, HP, Dell, DrayTek, SMC, Zyxel, Furukawa Electric, and many more
- Monitoring Systems: PRTG, Zabbix, Whatsup Gold, TheDude, RRDtoo
Always available for new projects! Contact me for any inquiries
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Identify the etlryl groups and the methyl groups in the structure of 4-sec-butyl-5ethyl-3- methyloctane, the compound discussed in Study problem 2.5. Note that these groups are not necessarily...
-
In a structure containing a pentacoordinate phosphorus atom, the bonds to three of the groups bound to phosphorus (called equatorial groups) lie in a plane containing the phosphorus atom (shaded in...
-
In the structure of 4- isopropy 1-2,4,5-trimethylheptane (Problem 2.9) (a) Identify the primary, secondary, tertiary, and quaternary carbons. (b) Identify the primary, secondary, and tertiary...
-
Three brothers, Daniel, David and Derrick have been discussing their respective taxation affairs and how much they dislike paying tax. None of them are Scottish taxpayers. Daniel's income for tax...
-
A 1.00-mg sample of substance of atomic mass 59.934 u emits particles with an activity of 1.131 Ci. Find the decay constant for this substance in s 1 and its half-life in years.
-
Dynabase Tool has forecast its total funds requirements for the coming year as shown in the following table. a. Divide the firm's monthly funds requirement into (1) A permanent component and (2) A...
-
Let \(Z\) be an \(\mathbb{R}\)-valued semi-martingale such that \(Z\) and \(Z_{-}\) do not vanish. Prove that \[ \mathcal{L}(Z)_{t}=\ln \left(\left|\frac{Z_{t}}{Z_{0}} ight| ight)+\int_{0}^{t}...
-
Wright Company?s balance sheet at December 31, 2011, is presented below. During January 2012, the following transactions occurred. (Wright Company uses the perpetual inventory system.)1. Wright paid...
-
For this requirement only, assume that at the end of December 2022 (before closing the books for the year), the Company reassesses the useful life of its "Machinery and Equipment". Originally, they...
-
Given the pK a values of methyl phosphate shown in this section, calculate the percentage of the un-ionized form, the monoanion form, and the di-anion form at pH 7.4.
-
Draw a structure for each of the following thioesters: (a) Cyclohexyl thiobenzoate (b) S-isopropyl butanethioate (c) S-phenyl cyclohexanecarbothioate
-
Ashcroft Financial paid $500,000 for a 30% investment in the common stock of Magic, Inc. For the first year, Magic reported net income of $220,000 and at year-end declared and paid cash dividends of...
-
If the functional currency of a foreign subsidiary is the local currency, is there any translation adjustment arising from goodwill on acquisition of a foreign subsidiary? Explain.
-
How would you know if people are really hearing what you want them to hear?
-
In recent decades, the Federal Reserve has transitioned from an intentionally vague and bureaucratic communication style to a more direct and transparent approach. What is the impact on inflation...
-
What dimensions of the Ignatian ethos resonate with your approach to leading organizational change during these turbulent times?
-
What happens to the amount Americans pay for imported goods if the price of the U.S. dollar depreciates? Will Americans import more or fewer goods if the U.S. dollar depreciates?
-
Suppose a random sample of size 36 is drawn from a population with a mean of 278. If 86% of the time the sample mean is less than 280, what is the population standard deviation? Explain.
-
Vectors are drawn from the center of a regular n-sided polygon in the plane to the vertices of the polygon. Show that the sum of the vectors is zero.
-
Explain how you would distinguish the compounds within each set by a simple chemical test with readily observable results, such as solubility in acid or base, evolution of a gas, and so forth....
-
(a) Give the structure of cocaine (Fig. 23.4) as it would exist in 1 M aqueous HCI solution. (b) What products would form if cocaine were treated with an excess of aqueous NaOH and heat? (c) What...
-
How would the basicity of trifluralin, a widely used herbicide, compare with that of N,N-diethylaniline: much greater, about the same, or much less? Explain Et Et O,N NO CF trifluralin
-
Two wires are tied to the 400 g sphere shown in (Figure 1). The sphere revolves in a horizontal circle at a constant speed of 7.3 m/s. A) What is the tension in the upper wire? B) What is the tension...
-
A small family contains four 10 W lamps, utilized 4 hours per day, two 60W fans, utilized 6 hours per day, and one refrigerator of 200W runs 20 hours daily. Then how many daily energy units are...
-
A 10.0 g object moving to the right at 19.0 cm/s makes an elastic head-on collision with a 15.0 g object moving in the opposite direction at 35.0 cm/s. Find the velocity of each object after the...

Study smarter with the SolutionInn App


