Keeping in mind that many intramolecular reactions that form six-membered rings are faster than competing intermolecular reactions
Question:
Keeping in mind that many intramolecular reactions that form six-membered rings are faster than competing intermolecular reactions (Sec. 11.8), predict the product of the reaction given in Fig. P11.56.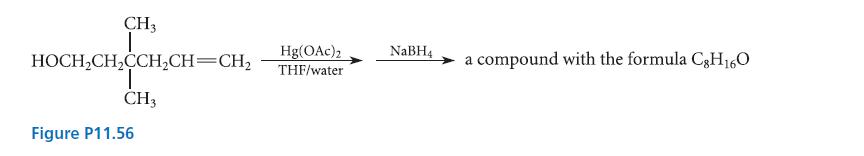
Transcribed Image Text:
CH3 I HOCH₂CH₂CCH₂CH=CH₂ CH3 Figure P11.56 Hg(OAc)2 THF/water NaBH4 compound with the formula C8H₁6O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The formula of the product indicates one degree of unsaturation Because the double bond under...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Required a. Use professional judgment in deciding on the preliminary judgment about materiality for earnings, current assets, current liabilities, and total assets. Your conclusions should be stated...
-
Keeping in mind that the open-ocean waters are highly oxic, predict the possible metabolic lifestyles of open-ocean Archaea and Bacteria. Why might rhodopsin-like pigments be more abundant in one...
-
Keeping in mind that aqueous Cu2+ is blue and aqueous Zn2+ is colorless, predict what you would observe over a severalday period if you performed the following experiments. a. A strip of Zn is placed...
-
How well employees modify their thoughts and behavior to align with and support a new or changing environment is known as Multiple Choice proactive task performance. proficient task performance....
-
How are manufacturing costs classified?
-
Bessies Quilting Company reported the following amounts on its balance sheet at December 31, 2010: Cash............... $ 5,000 Accounts receivable, net...... 40,000 Inventory............ 35,000...
-
For the following products and countries, identify the type of warehouse that should be used as well as the method of transportation that should deliver the product to end users. Using the Internet,...
-
Baker Mfg Inc. wishes to compare its inventory turnover to those of industry leaders, who have turnover of about 13 times per year and 8% of their assets invested in inventory. a. What is Bakers...
-
A wheel initially at rest begins rotating because of a constant angular acceleration. During a certain interval AT = 13s, the wheel goes through 84 rev and reaches an angular speed of 11 rev/s. (a)...
-
You are a manager for a company, Weighty Matters, that specializes in the manufacture of organic compounds containing 18 O, a heavy isotope of oxygen. You have assigned the task of preparing ether B...
-
For each of the following alkenes, state whether a reaction with OsO 4 followed by aqueous NaHSO 3 will give a racemic mixture of products that can (in principle) be resolved into enantiomers under...
-
At a recent conference on financial accounting and reporting, three participants provided examples of similar accounting changes that they had encountered in the last few months. They all involved...
-
Abarbanell, Inc., issues \(\$ 450,000\) of zero coupon bonds that mature in 10 years. Compute the bond issue price assuming that the bonds' market rate is: a. \(8 \%\) per year compounded...
-
What are the key contextual changes in B2B buyer behavior? What impact do these have on traditional price management practices?
-
Use the Brand24 case study to outline three to five critical success factors important in the price realization project. Why do you think theyre essential?
-
Suppose you estimate a regression using 20 observations and 16 independent variables. You compute R 2 to be .98. Explain why R 2 may not be an appropriate measure of the goodness of fit. Can you...
-
Smithe Inc. signed a 90-day, 6\% note payable for \(\$ 6,900\) on December 16 . Use the financial statement effects template to illustrate the year-end December 31 accounting adjustment Smithe must...
-
Explain the hybrid method of accounting.
-
If the cylinder described in Problem 21.3 were initially heated to 500F, how long would it take for the center of the cylinder to cool to 240F if it were constructed of a. Copper? b. Brass? c. Nickel?
-
Show how you could use the acetamidomalonate method to prepare the following amino acids: (a) Leucine (b) Tryptophan
-
Show how you could prepare the following amino acids using a reductive amination: (a) Methionine (b) Isoleucine
-
Show how you could prepare the following amino acids enantioselectively: (a) Pro (b) Val
-
Which answer choice does not represent preferred usage? We signed a 12-month lease agreement with the owner. Richard and Ann were able to secure a 25-year mortgage. The device has a 30-day warranty....
-
Robert has provided his personal information. Complete each statement to explain what information he has given. 1. canadienne: C'est sa enter answer . 2. G1K7X2: C'est son enter answer . 3. Robert:...
-
Eudora transfer the balance of $6400 to a new credit card at the beginning of the year. The card offered an introductory APR of 7.8% for the first three months and a standard APR of 26.5% thereafter....

Study smarter with the SolutionInn App


