Label each of the following molecules as a hydrogen-bond acceptor, donor, or both. Indicate the hydrogen that
Question:
Label each of the following molecules as a hydrogen-bond acceptor, donor, or both. Indicate the hydrogen that is donated or the atom that serves as the hydrogen-bond acceptor.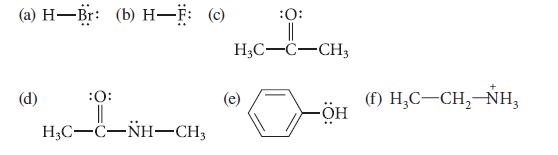
Transcribed Image Text:
(a) H-Br: (b) H-F: (c) (d) :0: HỌC–C–NH–CH, :0: || H3C-C-CH3 -ÖH (f) H₂C-CH₂-NH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a HBr is primarily a hydrogenbond donor because it is a strong acid a neutralBr ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Label each of the following molecules as a hydrogen-bond acceptor, donor, or both. Tndicate the hydrogen that is donated or the atom that serves as the hydrogen-bond acceptor. (a) (b) (c) H3c -...
-
Indicate whether each functional group of the five heterocyclic bases in nucleic acids can function as a hydrogen bond acceptor (A), a hydrogen bond donor (D), or both (D/A).
-
Label each of the following substances as an acid, base, salt, or none of the above. Indicate whether the substance exists in aqueous solution entirely in molecular form, entirely as ions, or as a...
-
______________ is an approach to doing business that attempts to maximize an organization's competitiveness through the continual improvement of the quality of its products, services, people,...
-
1. How did the Customs Service decide the nature of the Calcu-Folios in this case? 2. Why didnt the court agree with that classification?
-
Determine the total allowable 2010 earned income credit in each of the following situations: a. Rina is single and earns $6,300 in salary for the year. In addition, she receives $1,450 in...
-
Make decisions in the situations described in the Ethical Behavior Worksheet. You will not have all the background information on each situation; instead, you should make whatever assumptions you...
-
Scully Corporations comparative balance sheets are presented below. Additional information: 1. Net income was $22,630. Dividends declared and paid were $19,500. 2. All other changes in noncurrent...
-
Fill in the amounts (numbers) for each asset, liability, and equity category. Be sure to enter commas as needed in the numbers. For each entry labeled "total," you will need to do the math (addition)...
-
Within each set, arrange the compounds in order of increasing boiling point. (a) 4-ethylheptane, 2-bromopropane, 4-ethyloctane (b) 1-butanol, 1-pentene, chloromethane
-
Octane and 2,2,3,3-tetramethylbutane have boiling points that differ by about 20C (106C and 126C). Which has the higher boiling point and why?
-
For the following exercises, write the vector shown in component form.
-
How does AI relates to Accounting minor. 1. What is your major, and what is the importance of the information topic? a (Essentially, this part talks about how important your topic is to your major -...
-
Larry's friend, Leon, has decided to buy shares in Larry's new coffee shop, Latte Larry's. The current market price of the stock is $20. Over the next year, Leon expects the company to pay dividends...
-
Long-term debt ratio 0.3 Times interest earned 10.0 Current ratio 1.6 Quick ratio. 1.0 Cash ratio 0.3. Inventory turnover 3.0 Average collection period 73 days Use the above information from the...
-
Joella is out shopping and finds a pair of sneakers originally valued at $96.00. They are now on sale for 75% of the original price. What is the new cost of the item? 2 answers
-
Assume that you are analyzing the financial statements of Telstra for the year 2022. Where can you find the information to examine its performance? How would you comment on the performance of Telstra...
-
Do you think the overall level of R&D would increase or decrease over the next 20 to 30 years if the lengths of new patents were extended from 20 years to, say, forever? What if the duration were...
-
Time Solutions, Inc. is an employment services firm that places both temporary and permanent workers with a variety of clients. Temporary placements account for 70% of Time Solutions' revenue;...
-
A solution of a 0.0014 g of benzophenone in 1 L of ethanol has A = 0.153 (1 cm cell) at ? max = 252 nm. Calculate the molar absorptivity of benzophenone. Benzophenone
-
Trans-1-Phenyl-1, 3-butadiene has max = 280 ( = 27,000) calculate the concentration of a solution that has A = 0.643 at 280nm in a 1 cm cell.
-
Nitro methane max = 275nm ( = 1.5) what kind of transition is responsible for this absorption?
-
An aluminium object with a mass of 2.13 kg and at a temperature of 29.8 C comes to thermal contact with a 7.65 kg copper object which is initially at a temperature of 99.1 C. What is going to be the...
-
y (m) t=0 seconds 20 -20 4 Shown below is a transverse wave, moving to the right (positive x-direction). The period of oscillation is 6 seconds. (The arrow is there to help you see the motion of the...
-
A 1.95-kg particle has a velocity (2.04 - 3.09 ) m/s, and a 3.02-kg particle has a velocity (1.09 +6.03 j) m/s. (a) Find the velocity of the center of mass. i+ 1) m/s (b) Find the total momentum of...

Study smarter with the SolutionInn App


