Only one of the following three alkyl halides can be prepared as the major product of the
Question:
Only one of the following three alkyl halides can be prepared as the major product of the addition of HBr to an alkene. Which compound can be prepared in this way? Explain why the other two cannot be prepared in this way.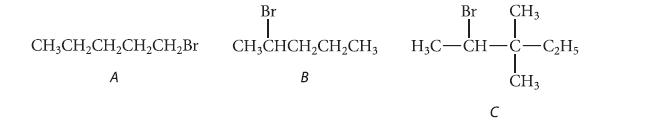
Transcribed Image Text:
CH3CH₂CH₂CH₂CH₂Br A Br I CHỊCHCH,CH,CH, B Br I CH3 H3C-CH-C-C₂H5 I CH3 C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
If these alkyl halides could be prepared from alkenes the alkene sta...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1. Hannah is applying for a life policy on her girlfriend Sarahs life. The policy is $500,000 and carries a large premium. Hannah is the main earner, so she is concerned about not being able to pay...
-
MCQ questions: 1. An increase in oil prices, such as the oil shocks in the 70s, lead to _______ thereby causing ________ a movement along the AS curve; cost-push inflation a leftward shift in the AS...
-
Four million infants in the United States consume 80 million cases of jarred baby food annually, representing a domestic market of $865 million to $1 billion. The baby food market is dominated by...
-
What is the Antebellum Interregional growth hypothesis?
-
Was the phrase defamatory, or was it deliberate exaggeration that no reasonable person would take literally?
-
Leonard and Michelle have asked you to prepare their statement of changes in net worth for the year ended August 31, 20X3. They have prepared the following comparative statement of financial...
-
Julie Palmer has trouble keeping her debits and credits equal. During a recent month, Julie made the following errors: a. Julie recorded a $750 payment of rent by debiting rent expense for $75 and...
-
Learning curve, incremental unit-time learning model. Learning curve, incremental unit-time learning model, Assume the same information for Global Defense as in Exercise 10-29, except that Global...
-
Compare price risk (interest rate risk) and reinvestment risk in relation to bonds?
-
Draw curved-arrow mechanisms and transition-state structures for each of the following two reactions. Each reaction occurs as a single step. (a) CHCH-Br: + FCH3 CHCH-CH3 + :Br: (b) (CH3)3C-Br:...
-
Which of the following carbocations is likely to rearrange? If rearrangement occurs, give the structures of the rearranged carbocations. @ H I + CH3 (b) CH3 CH3 T T CH3CH-C-CH3 + O
-
Under what circumstances would you include more than one index in a lengthy report?
-
ces The contribution format income statement for Huerra Company for last year is given below: Sales Variable expenses Contribution margin Fixed expenses Net operating income Income taxes @ 40% Net...
-
(2) Electric force between two positive charges Drag the sensor to all the locations marked with "x" in the following graph - Figure 2. For each location, (a) draw the red arrow in proper direction...
-
Streaming of YouTube videos became increasing popular as the costs of cable and satellite television have surged over the last 10 years. For instance, during the coronavirus pandemic, a YouTube...
-
Create a Project Roadmap that discusses the company your team will study. Include a brief overview of the current issue summarized with a short and succinct problem statement, no more than two short...
-
Explain the induced fit model and contrast it with the lock-and-key model in the context of enzyme-substrate interactions .
-
Ten major college football bowl games were played in January 2010, with the University of Alabama beating the University of Texas 37 to 21 to become the national champion of college football. The...
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
Give IUPAC names for the followingcompounds: (b) (a) The three isomers of C5H12 CH CH;CH-CHH CH (d) (c) CH CH (CH)2CHCH2CH3 (CH3)3CH-H2C CH
-
Draw structures corresponding to the following IUPAC names: (a) 3, 4-Dimethylnonane (b) 3-Ethyl-4, 4-dimethylheptane (c) 2, 2-Dimethyl-4-propylocatane (d) 2, 2, 4-Trimethylpentane
-
Name the eight 5-carbon alkyl groups you drew in Problem 3.7.
-
Helium is pumped into a spherical balloon at a rate of 3 cubic feet per second. How fast is the radius increasing after 3 minutes? Note: The volume of a sphere is given by V = (4/3). Rate of change...
-
Assume that x and y are both differentiable functions of t. Find the required values of dy dx and dt dt x+y2= = 169 dy (a) Find given x = 5, y = 12, and dx dt = 6. dt dy x dt dx (b) Find given x 12,...
-
Suppose that f() = -7 and f'(): = 4, and let g(x) = f(x) sin x and h(x) 1. Find g'(/3). Answer: g'(/3) = 2. Find h'(/3). Answer: h'(/3) = COS X = -. Answer the following questions. f(x)

Study smarter with the SolutionInn App


