Outline a Williamson ether synthesis for tert-butyl methyl ether. CH3 H3C-C-0-CH3 I CH3 tert-butyl methyl ether
Question:
Outline a Williamson ether synthesis for tert-butyl methyl ether.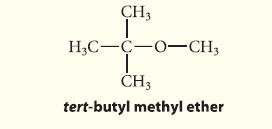
Transcribed Image Text:
CH3 H3C-C-0-CH3 I CH3 tert-butyl methyl ether
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
From Eq 116 two possibilities for preparing this compound are the reaction ...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A good Williamson synthesis of ethyl methyl ether would be What is wrong with the following proposed synthesis of ethyl methyl ether? First, ethanol is treated with acid to protonate the hydroxyl...
-
Show how you would use the Williamson ether synthesis to prepare the following ethers. You may use any alcohols or phenols as your organic starting materials. (a) cyclohexyl propyl ether (b)...
-
(a) Outline two methods for preparing isopropyl methyl ether by a Williamson ether synthesis. (b) One method gives a much better yield of the ether than the other. Explain which is the better method...
-
MOSS COMPANY Income Statement \ table [ [ , ] , [ Sales For Year Ended December 3 1 , 2 0 2 1 , $ 5 2 5 , 0 0 0 es MOSS COMPANY Income Statement For Year Ended December 31, 2021 Sales Cost of goods...
-
Buckaroo Company and Yearling Company entered into the following transactions: a. Buckaroo Company sold merchandise to Yearling Company for $135,000, terms 2/10, n/30. b. Prior to payment, Yearling...
-
Use Microsoft Access or similar database software to create a DBMS for the imaginary company called TopText Publishing, which is described in Case In Point 9.1 on page 365. Add several sample records...
-
Determine if the mixed method aspect is concurrent, sequential,, or nested. Develop a hypothetical research scenario that would necessitate the use of the Mixed Method A-B-A Design. The research will...
-
A comparative balance sheet for Orozco Corporation is presented below. Additional information:1. Net income for 2012 was $105,000.2. Cash dividends of $40,000 were declared and paid.3. Bonds payable...
-
Calculate the following for a 3 ft 4 ft window with the label to the right. The summer design conditions for peak cooling load are 70 and 60% RH inside; 90 and 90% outside; and a peak direct solar...
-
Arrange the ions in the following list in order of increasing acidity, and explain your reasoning. | A H3C B CH3 H3C C H D
-
(a) Describe the bond for the double bond in Ph 3 P=CH 2 ; that is, what orbitals are involved on carbon and phosphorus? (b) Draw a resonance structure for the compound in part (a) that maintains...
-
Plot each point in a rectangular coordinate system. (3, -3)
-
It is July 2016. A mining company has just discovered a small deposit of gold. It will take six months to construct the mine. The gold will then be extracted on a more or less continuous basis for...
-
What is arbitrage? Explain the arbitrage opportunity when the price of a dually listed mining company stock is $50 (USD) on the New York Stock Exchange and $60 (CAD) on the Toronto Stock Exchange....
-
The spot price of silver is \(\$ 15\) per ounce. The storage costs are \(\$ 0.24\) per ounce per year payable quarterly in advance. Assuming that interest rates are \(10 \%\) per annum for all...
-
The 2-month interest rates in Switzerland and the United States are, respectively, \(2 \%\) and \(5 \%\) per annum with continuous compounding. The spot price of the Swiss franc is \(\$ 0.8000\). The...
-
Alta Ltd., which sells computers, had the following receivables and payables: cash sales of 50 percent; credit sales of 50 percent with terms of payment 15-20 days after sales; inventory purchase all...
-
The Miller family has owned several large apartment buildings for many years. They organized Miller Properties, Inc., to own and manage the properties. The corporation is an accrual basis taxpayer...
-
Use the formula to determine the value of the indicated variable for the values given. Use a calculator when one is needed. When necessary, use the key on your calculator and round answers to the...
-
Show the steps involved in a laboratory synthesis of the DNA fragment with the sequence CTAG.
-
The final step in DNA synthesis is deprotection by treatment with aqueous ammonia. Show the mechanisms by which deprotection occur at the points indicated in the followingstructure: . DMTO N. 'N'...
-
Draw the structure of cyclic adenosine mono phosphate (cAMP), a messenger involved in the regulation of glucose production in the body. Cyclic AMP has a phosphate ring connecting the 3' and 5'...
-
The value of a business's fixed asset life ratio has increased in each of the last three years. What conclusion could you draw from this?
-
For the first 10 days of one of his 30 day billing cycles his balance was $2500 either made a payment for $1600 so his balance decreased 908 remains that amount for the next 10 days Armando that made...
-
For best collection results, collection agencies like to get the account at a maximum of: a. 1 or 2 months after the debt has occurred. b. 2 or 3 months after the debt has occurred. c. 3 or 4 months...

Study smarter with the SolutionInn App


