Outline a Wittig synthesis for each of the following alkenes; give two Wittig syntheses of the compound
Question:
Outline a Wittig synthesis for each of the following alkenes; give two Wittig syntheses of the compound in part (a).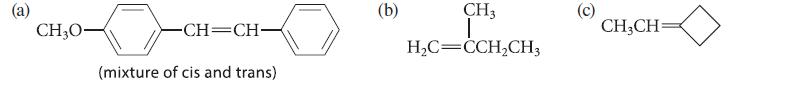
Transcribed Image Text:
CH₂0- -CH=CH- (mixture of cis and trans) (b) CH3 I H₂C=CCH₂CH3 CH₂CH=
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Either half of the alkene can in principle be derived ...View the full answer

Answered By

Shem Ongek
I am a professional who has the highest levels of self-motivation. Additionally, I am always angled at ensuring that my clients get the best of the quality work possible within the deadline. Additionally, I write high quality business papers, generate quality feedback with more focus being on the accounting analysis. I additionally have helped various students here in the past with their research papers which made them move from the C grade to an A-grade. You can trust me 100% with your work and for sure I will handle your papers as if it were my assignment. That is the kind of professionalism that I swore to operate within. I think when rating the quality of my work, 98% of the students I work for always come back with more work which therefore makes me to be just the right person to handle your paper.
4.80+
174+ Reviews
426+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Outline practical syntheses of each of the following compounds from alcohols containing no more than four carbon atoms and any necessary organic or inorganic reagents. In many cases the desired...
-
Outline practical syntheses of each of the following compounds from alcohols containing no more than four carbon atoms and any necessary organic or inorganic reagents. In many cases the desired...
-
Starting with an appropriate alkyl halide and base, outline syntheses that would yield each of the following alkenes as the major (or only) product: (a) (b) (c) (d) (e)
-
The dehydration butanol of alumina is carried out over a silica-alumina catalyst at 680K. CH3CH2CH2CH20H------->cat CH3CH=CHCH3 + H2O The rate law is -r Bu = KPBU/(1+KBuPBul with k= 0.054...
-
Amazon.comlike all other businessesmakes adjusting entries prior to year-end in order to measure assets, liabilities, revenues, and expenses properly. Examine Amazons balance sheet and Note 3. Pay...
-
Identify the possible values of the following random variables. Which of the random variables are discrete? a. The numerical grade a student receives in a course. b. The grade point average of a...
-
Hooters Restaurant in Myrtle Beach, South Carolina, used an alternative dispute resolution program, a program to resolve disputes outside the traditional court system. Employees of Hooters had to...
-
A company processes a chemical, DX-1, through a pressure treatment operation. The complete process has two outputs, L and T. The January costs to process DX-1 are $50,000 for materials and $100,000...
-
The interest rate is fixed at 10%. You are offered an asset that pays $1,000 from now until eternity at yearly intervals. You find its present value by calculating the sum of the discounted annual...
-
Give the structure of an aldehyde C 8 H 8 O 2 that would be oxidized to terephthalic acid by KMnO 4 . HO-C- O -C-OH terephthalic acid
-
Outline a synthesis of 1,4-dimethoxy-2-propylbenzene from hydroquinone (p-hydroxyphenol) and any other reagents.
-
1. Research the FTC's deceptive advertising policy and report on another case involving substantiation of specific claims. 2. The advertising industry has established the National Advertising...
-
Find the area of the irregular quadrilateral (show your work plz) 91.98' (RM) RAD=410.00' (R) 5' P.U. D.E. OT 117 NOI 03'50"In 67.85' (R/M) 10' P.U. & D.E. FENCE FJ.P. 3/4" FENCE 0.20 SOUTH 0.40 EAST...
-
I need the answer as soon as possible Hiw 20.55. 2040 2021. 2025 2055. Estimate the increase in the population. Suppose the number from 1940 to 1998. As for the population in these years, we put it,...
-
8.Define and describe each of the contingency effectiveness approaches. 9.When would you recommend that the resource-based approach be used to gauge organizational effectiveness? 10.Explain why...
-
What does evidence from the human development index suggest about changes in the quality of life over the last 120 years? (20 marks) This question is related to economic history. Please go into...
-
Can you please help me make sense of these questions? I dont understand left side versus right side, please help! Q1: Why are the calculated price elasticity of demand results not the same along the...
-
Ontario, Inc. manufactures two products, Standard and Enhanced, and applies overhead on the basis of direct-labor hours. Anticipated overhead and direct-labor time for the upcoming accounting period...
-
Explain five different cases of income exempt from tax with clear examples.
-
Experiments show that the molecular weight of acetic acid in the vapor state (just above its boiling point) is approximately 120. Explain the discrepancy between this experimental value and the true...
-
Amides are weaker bases than corresponding amines. For example, most water-insoluble amines (RNH2) will dissolve in dilute aqueous acids (aqueous HCl, H2SO4, etc.) by forming water-soluble...
-
While amides are much less basic than amines, they are much stronger acids. Amides have pKa values in the range 14-16, whereas for amines, pKa = 33-35. (a) What factor accounts for the much greater...
-
Draw a glass box analysis for a basic screw driver whose use is to change rotational motion into linear motion in order insert a screw into something. Inputs should cover the following information:...
-
Beginning Inventory consists of 4 items at $10 each. During the month, the company purchased 3 items for $11 each and it sold 3 items. Using last-in, first-out, the 3 goods sold are ______. Multiple...
-
a) Calculate and plot the air specific impulse pair =T/(9) and specific fuel consumption SFC=9/T as a function of the stagnation pressure ratio for an ideal turbojet engine. Assume the flight Mach...

Study smarter with the SolutionInn App


