The following triester is a powerful explosive, but is also a medication for angina pectoris (chest pain).
Question:
The following triester is a powerful explosive, but is also a medication for angina pectoris (chest pain). From what inorganic acid and what alcohol is it derived?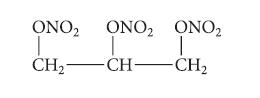
Transcribed Image Text:
ONO₂ T CH₂- ONO₂ ONO₂ L I -CH-CH₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
This is a triester of 123propanetriol glycerol and nit...View the full answer

Answered By

Akash Goel
I am in the teaching field since 2008 when i was enrolled myself in chartered accountants course
Since then i have an experience of teaching of class XI, XII, BCOM, MCOM, MBA, CA CPT.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
First Aid and Emergencies An emergency is defined as a situation where there is an immediate risk to health, life, property or environment and urgent action is needed to try to stop the situation...
-
Nitroglycerine, C 3 H 5 (NO 3 ) 3 (), is an explosive most often used in mine or quarry blasting. It is a powerful explosive because four gases (N 2 , O 2 , CO 2 , and steam) are formed when...
-
The following study is described in Chinchilli, Schwab, and Sen (1989). The pain of angina is caused by a deficit in oxygen supply to the heart. Calcium channel blockers like verapamil will dilate...
-
Write code in MATLAB (Radionuclide) (half-life) U-238 4.468 x 10 years U-235 703.8 x 10 years Mo-99 67 hours Tc-99m 6.04 hours Given the formula, where is the decay constant used in , calculate the...
-
Molly Maloney is an employee of Marshall Company, a small manufacturing concern. Her responsibilities include opening the daily mail, depositing the cash and checks received into the bank, and making...
-
On December 31, Year 8, U.S. Dental Supplies (USDS) created a wholly owned foreign subsidiary, Funi, Inc. (FI), located in the country of Lumbaria. The condensed balance sheet of Funi as of December...
-
By using the five principles of regulatory design outlined in this chapter, consider how ecological reporting to external stakeholders provides a fundamental foundation for regulation of...
-
Carla Carpet manufactures broadloom carpet in seven processes: spinning, dyeing, plying, spooling, tufting, latexing, and shearing. In the Dyeing Department, direct materials (dye) are added at the...
-
Where would a manager find all the costs associated with a particular job or project, including materials, labor, and overhead costs?
-
Give the product expected, if any, when 1-butanol (or other compound indicated) reacts with each of the following reagents. (a) Concentrated aqueous HBr, H 2 SO 4 catalyst, heat (b) Cold aqueous H 2...
-
(a) How many electrons are involved in the oxidation of triphenylphosphine (Ph 3 P;) to triphenylphosphine oxide (Ph 3 P = O)? Show your reasoning. (b) Draw a resonance structure for...
-
Greg Failla owns Showbiz Video. He uses the following accounts to record business transactions. For each transaction: 1. Prepare a T account for each account listed above. 2. Analyze and record each...
-
The first professional football player was W. W. Pudge Heffelfinger, a former All-American at Yale, who was working on a railroad in 1892 when he was paid $500 to play one game for the Allegheny...
-
Under a single-rate electricity tariff for households and small businesses in Australia, a flat usage rate is charged for "blocks" of electricity consumed. The rate for the first block is higher than...
-
Suppose a certain battery has an internal emf of \(9.00 \mathrm{~V}\) but the potential difference across its terminals is only \(85.0 \%\) of that value. If that battery is connected to a \(56.0-\mu...
-
A bar magnet is enclosed by a spherical surface. (a) What is the magnetic flux through the entire surface? (b) If the magnetic flux through the hemisphere closest to the magnet's north pole is...
-
Find the state-space form directly from the I/O equation. \(3 \dddot{y}+\dot{y}+2 y=3 \ddot{u}+2 u\)
-
Palmer Company uses an activity-based costing system. It has the following manufacturing activity areas; related drivers used as allocation bases, and cost allocation rates: During the month, 100...
-
Find a polar equation for the curve represented by the given Cartesian equation. 4y 2 = x
-
Is the pro-R or pro-S hydrogen removed from citrate during the dehydration in step 2 of the citric acid cycle? Does the elimination reaction occur with syn or antigeometry? " 02C. "02c. .co, CO2...
-
Write a mechanism for step 6 of gluconeogenesis, the reduction of 3-phospho-glyceryl phosphate with NADH/H+ to yield glyceraldehydes 3-phosphate.
-
Write all the steps in the transamination reaction of PMP with -ketoglutarate plus a lysine residue in the enzyme to give the PLPenzyme imine plus glutamate.
-
Vigo Vacations has $300 million in total assets, $7 million in notes payable, and $44 million in long- term debt. What is the debt ratio? Do not round intermediate calculations. Round your answer to...
-
Fill out the daily spending diary for a 51-year-old woman with two kids, spending $1600 on rent, not eating out frequently, purchasing groceries, and having an electric car to minimize gas expenses....
-
Smiling Elephant, Inc., has an issue of preferred stockoutstanding that pays a $5.60 dividend every year, inperpetuity. If this issue currently sells for $80.40 per share, what is therequired return?...

Study smarter with the SolutionInn App


