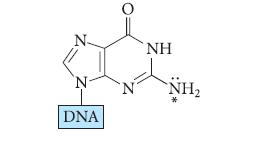
The group in DNA that reacts with the diol-epoxide has the following general structure. Using mechanistic reasoning,
Question:
The group in DNA that reacts with the diol-epoxide has the following general structure. Using mechanistic reasoning, show how the amino group indicated by the asterisk (*) might react with the epoxide group of the diol-epoxide in Eq. 16.50.![benzo[a]pyrene living tissue (enzymes, O) HO" OH benzo[a]pyrene diol-epoxide (16.50)](https://dsd5zvtm8ll6.cloudfront.net/images/question_images/1701/6/6/6/340656d5e24769351701666338943.jpg)
Transcribed Image Text:
benzo[a]pyrene living tissue (enzymes, O₂) HO" OH benzo[a]pyrene diol-epoxide (16.50)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
The nucleophilic nitrogen adds to the less stericallyh...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Proteins are synthesized with a particular amino acid sequence through the translation of information encoded in messenger RNA by an RNAprotein complex called a ribosome. Amino acids are specified by...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Describe several features of the ACA that are designed to minimize adverse selection. Why might some of those measures not end up working very well?
-
Interpreting common-size income statements Thales Group (Thales is a French electronics firm that specializes in information systems. aerospace, and defense Thales applies IFRS and reports its...
-
Windy Corporation, which uses the allowance method, has accounts receivable of $50,800 and an allowance for uncollectible accounts of $9,800. An account receivable from Tom Novak of $4,400 is deemed...
-
A \(70-\mathrm{kg}\) skier is being towed on a rope behind a \(450-\mathrm{kg}\) snowmobile on a smooth, snow-covered surface at \(10 \mathrm{~m} / \mathrm{s}\) when the snowmobile hits a patch of...
-
Assume that you are the nursing administrator for a medical group that expects a severe outbreak of flu this winter. You hire additional staff to treat patients and administer shots. Your special...
-
Define a problem, problem identification, problem solving, and decision making and discuss types of decisions and decision-making conditions. Discuss the nature, and process of decision making
-
Using benzene and any other reagents, outline a synthesis of each of the following compounds. (a) Cyclohexylcyclohexane (b) Tert-butylcyclohexane
-
In each of the following sets, rank the compounds in order of increasing harshness of the reaction conditions required to accomplish the indicated reaction. (a) Sulfonation of benzene, m-xylene, or...
-
Managements responsibility for the financial statements includes: (a) Selecting internal controls tests. (b) Selecting samples for audit testing. (c) Selecting and applying appropriate accounting...
-
Discuss the four areas of fact-based decision making. In your opinion, what is the greatest obstacle to using fact-based decision making?
-
How would you determine if a potential partnership aligns with your project vision?
-
A key supplier for your project has not been returning your calls or responding to your e-mails. This is an example of a(n) ________________, which indicates that a risk is likely to occur.
-
You are the project manager of a construction project for a large organization and will be relying mostly on independent contractors to execute the project work. Which type of contract would you...
-
What is the purpose of identifying a threshold for project metrics?
-
Use the following data to construct a 99% confidence interval to estimate D. Assume the differences are normally distributed in thepopulation. Respondent eore After Before 47 63 35 36 56 38 50 39 27...
-
Will the prediction interval always be wider than the estimation interval for the same value of the independent variable? Briefly explain.
-
Although aldehydes and ketones are weak acids, their a-hydrogens are rnore than 30 pKa units more acidic than the hydrogen's of alkanes. Using polar effects and resonance effects in your argument,...
-
Which of the following two alkyl halides would react most rapidly in a solvolysis reaction by the SN1 mechanism? Explain your reasoning. Ch 1,0-CH-CH-CH,-Cl A (trans isomer) CH.O-C-CH,-CI CH2
-
Most alkyl bromides ile water-insoluble liquids. Yet, when 7-bromo- 1,3,5-cycloheptatriene was first isolated, its high melting point of 203oC and its water solubility led its discoverers to comment...
-
A solid of mass m , specific heat c, and temperature 195 o C is placed into a water of mass 4 m (and specific heat 1 cal/g o C) at 10 o C. The objects are allowed to come to equilibrium temperature...
-
The de voltage, Vdc, in Figure 1 represents the voltage generated by an array of solar cells constructed on rooftop of BN building. These solar panels are capable of producing 1.8 kW of green energy....
-
The magnitude of the electric field at a point P is 2.3 x 10^7 NIC. What is the magnitude of the electric force that would be exerted at P on (a) an electron? (b) an oxygen nucleus (charge +8e)?
Digital Marketing Analytics In Theory And In Practice 1st Edition - ISBN: 979-8853048072 - Free Book

Study smarter with the SolutionInn App


