The reaction of water with metaphosphate ion is shown in Eq. 25.19b. Could nitrate ion undergo an
Question:
The reaction of water with metaphosphate ion is shown in Eq. 25.19b. Could nitrate ion undergo an analogous reaction with aqueous NaOH? If so, draw the structure of the product. If not, explain why.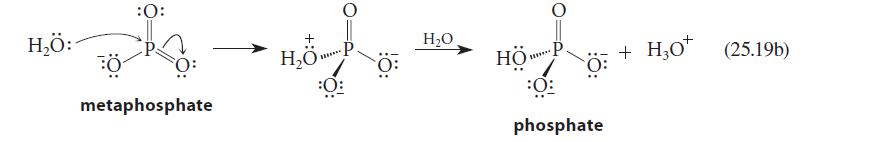
Transcribed Image Text:
H₂Ö: :O: metaphosphate H₂O P ***** :0 H₂O HÖP. ******* phosphate H30* (25.19b) + H₂0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Nitrate ion is stabilized by resonance which distributes the negative charge ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The reaction of water with CH3Cl in acetone as a solvent is represented by the equation CH3Cl H2O CH3OH + HCl The rate of the reaction doubles when the concentration of CH3Cl is doubled and it...
-
Cocaine metabolism in rats can be studied by injecting the drug and periodically withdrawing blood to measure levels of metabolites by HPLC-mass spectrometry. For quantitative analysis, isotopically...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
In the year to 5 April 2021, Thomas More made the following disposals: (i) A flat in a house that he had purchased on 1 December 2010 for 80,000. It had never been occupied as the main residence and...
-
The isotope 24 Na is a emitter with a half-life of 15 h. A saline solution containing this radioactive isotope with an activity of 600 kBq is injected into the bloodstream of a patient. Ten hours...
-
Strong Tool Company has been considering purchasing a new lathe to replace a fully depreciated lathe that will last 5 more years. The new lathe is expected to have a 5-year life and depreciation...
-
Review the Ethical Focus feature, Whats in a Name? Youre Hired... or Not!, and use Bruners model of the perceptual process to explain why job applicants with ethnic-sounding names are less likely to...
-
A review of the ledger of Terrell Company at December 31, 2012, produces these data pertaining to the preparation of annual adjusting entries.1. Prepaid Insurance $15,200. The company has separate...
-
Read the Change Metaphor Entitled Who Moved My Cheese and Critically Reflect on the Following Points 1) Being a metaphor in the story, what the Cheese represents in the real life change situation?...
-
The conjugation of alcohols containing nonpolar groups (for example, phenol) to glucuronic acids is a key process by which these alcohols can be transformed into, and excreted as, water-soluble...
-
Account with a mechanism for the fact that the hydrolysis of trimethyl phosphate to dimethyl phosphate in acidic solution containing 18 O-labeled water gives methanol containing 18 O and dimethyl...
-
What are the various ways the LCM determination can be made?
-
For each of the following institutions, provide a real-world example and explain how it promotes economic growth. a. Enforceable property rights b. Predictable and stable government c. Efficient...
-
What is the equilibrium unemployment rate and why isnt it equal to zero? Explain.
-
From your own experiences, provide an example of each: consumption, investment, government purchases, and net exports. Problem 7 Explain how total spending and total income for an economy must be...
-
Youve started developing an app that examines students personalities and other characteristics, and sorts them into highly effective study groups. Using the cost-benefit principle, compare your...
-
Why would a company want to pay its workers more than the prevailing market wage? What is the impact on employers, workers, and the overall labor market?
-
The Aluminum Association reports that the average American uses 56.8 pounds of aluminum in a year. A random sample of 51 households is monitored for one year to determine aluminum usage. If the...
-
In Exercises, find the equation of the tangent line at the given point on each curve. 2y 2 - x = 4; (16, 2)
-
Pentanoic acid and methyl butyrate are constitutional isomers. Which has the higher boiling point and why?
-
Klutz McFingers. a graduate student in his ninth year of study, has suggested the following synthetic procedures and has come to you in the hope that you can explain why none of them works very well...
-
Complete the reactions given in Fig. P21.52 by giving the principal organic products. Explain how you arrived at your answers. NaOH CH O (trace) H,C CCHO CH +CH OH (solvent) Ph NH2 1 (CgH,NO3)...
-
How can accountability be fostered in cross-border contexts and global governance frameworks, such as international treaties or multinational corporations, to address transnational challenges and...
-
According to the manufacturer of M&M candy, the color distribution for plain chocolate M&Ms is 13% brown, 13% red, 14% yellow, 24% blue, 20% orange, and 16% green. A 1.69-ounce bag of plain chocolate...
-
Discuss the amount of force that police can use in effectuating an arrest and preventing a suspect from escaping or overcoming resistance. Be sure to cite relevant case law bcj 3950 constitutional...

Study smarter with the SolutionInn App


