Using the Hckel 4n + 2 rule, determine whether each of the following compounds is likely to
Question:
Using the Hückel 4n + 2 rule, determine whether each of the following compounds is likely to be aromatic. Explain how you arrived at the π-electron count in each case.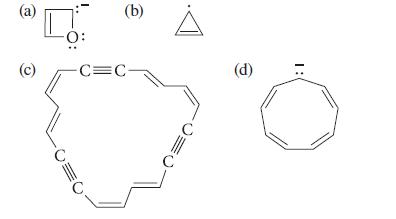
Transcribed Image Text:
(a) (c) CO (b) -C=C- (d) :1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b c d This species contains six 7 electrons two from the double bond two from ...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the values of the diffusion coefficients for the interdiffusion of carbon in both iron (BCC) and iron (FCC) at 900C Which is larger Explain why this is the case
-
Using the Huckel 4n + 2 rule, determine whether each of the following compounds is likely to be aromatic. Explain how you arrived at the -electron count in each case. (a) (b)
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Research is a process of discovering new knowledge. In the Code of Federal Regulations (45 CFR 46.102(d)) pertaining to the protection ofhuman subjects,research is defined as:...
-
What are the six articles of ethical conduct set out under section I of the AICPAs Code of Professional Conduct?
-
Bridgton Golf Academy is evaluating different golf practice equipment. The Dimple-Max equipment costs $94,000, has a three-year life, and costs $8,600 per year to operate. The relevant discount rate...
-
Mary Kay Morrow began working for Hallmark in 1982. At the beginning of 2002, Hallmark adopted the Hallmark Dispute Resolution Program, which required, among other things, that claims against the...
-
Eagle Carts, Inc., produces special-order golf carts, so Eagle Carts uses a job order costing system. Overhead is applied at the rate of 90 percent of direct labor cost. A list of transactions for...
-
How would you import a relational model design and generate a logical model from a relational model?
-
Give the principal product(s) expected, if any, when trans-1,3-pentadiene reacts under the following conditions. Assume one equivalent of each reagent reacts unless noted otherwise. (a) Br 2 (dark)...
-
The following molecule has a barrel shape (in which the benzene rings are the walls of the barrel). It forms a noncovalent complex with the iodide salt of acetylcholine in chloroform solvent....
-
Define the clausal form of formulas and Horn clauses.
-
Market price ____________equilibrium price. a) must always be equal to b) must always be above c) must always be below d) may be equal to
-
Haley Megan Fosnough-Biersmith goes shopping for shoes and has plenty of money with her. She will keep buying shoes until _______. a) her total utility equals the price b) her marginal utility equals...
-
Total cost is the sum of ____. a) marginal cost and fixed cost b) marginal cost and variable cost c) variable cost and fixed cost
-
Market price may not reach equilibrium if there are ______. a) both price ceilings and price floors b) neither price ceilings nor price floors c) only price ceilings d) only price fl oors
-
Which statement is true? a) A change in demand is the same thing as a change in the quantity demanded. b) The supply curve moves upward to the left. c) The law of demand is no longer valid. d) A rise...
-
A tornado extensively damaged the community in which Bodine Company had its primary manufacturing facilities. Bodine gives $1,000 to each household that suffered damage from the tornado to help...
-
Two mutually exclusive investment alternatives are being considered. Alternative A requires an initial investment of $20,000 in a machine. Annual operating and maintenance costs are anticipated to be...
-
How might you use IR spectroscopy to distinguish among the three isomers 1 -butyne, 1, 3-hutadiene, arid 2-butyne?
-
Would you expect two enantiomers such as (R)-2-brornobutane and S)-2-bromobutane to have identical or different IR spectra explain.
-
Would you expect two diastereomers such as meso-2, 3-dibromobutanc and (2R, 3R)-dibromo butane to have identical or different IR spectra? Explain.
-
A grocery store consumer market in a midwest city involves a competition for market shares among two grocery stores: Schnuks (Sch) and Meijers (Mjr). A consumer survey revealed that at the moment...
-
Income statements and balance sheets data for Virtual Gaming Systems are provided below. VIRTUAL GAMING SYSTEMS Income Statements For the years ended December 31 2022 2021 Net sales $ 3,550,000 $...
-
Tyler purchased a disability policy with a waiver of premium rider on April 1. He is disabled on May 1. On June 1, he receives proof of permanent and total disability, and submits a claim. He begins...

Study smarter with the SolutionInn App


