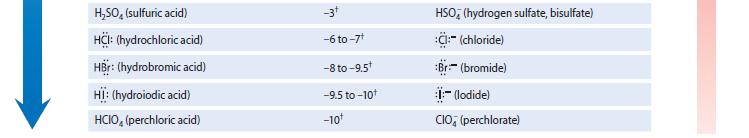
Using the pK a values in Table 3.1, calculate the equilibrium constant for each of the following
Question:
Using the pKa values in Table 3.1, calculate the equilibrium constant for each of the following reactions.
(a) NH3 acting as a base toward the acid HCN
(b) F– acting as a base toward the acid HCN
Transcribed Image Text:
GREATER ACIDITY TABLE 3.1 Relative Strengths of Some Acids and Bases Conjugate acid pk NH, (ammonia) RÖH (alcohol) HÖH (water) HPO (hydrogen phosphate) RSH (thiol) R₂NH (trialkylammonium ion) NH, (ammonium ion) HCN (hydrocyanic acid) H₂PO, (dihydrogen phosphate) HSH (hydrosulfuric acid) R-C-OH (carboxylic acid) HF: (hydrofluoric acid) H₂PO4 (phosphoric acid) HNO, (nitric acid) H₂O* (hydronium ion) H₂C- -SO₂H (p-toluene- sulfonic acid) ~35t 15-19 15.7 12.3 10-12* 9-11* 9.25 9.40 7.21 7.0 4-5* 3.2 2.2 -1.3 -1.7 -2.8+ Conjugate base -NH₂ (amide) RÖ:- (alkoxide) HÖ:- (hydroxide) PO (phosphate) RS (thiolate) R₂N: (trialkylamine) H₂N: (ammonia) -:CN (cyanide) HPO (hydrogen phosphate) HS:- (hydrosulfide) R-C-Ö: (carboxylate) F:-(fluoride) H₂PO (dihydrogen phosphate) NO; (nitrate) H₂O (water) H₂C- (p-toluene- -SO sulfonate, or "tosylate") GREATER BASI CITY
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a b The acidbase reaction is HCN NH3 CN NH4 When NH3 ...View the full answer

Answered By

Shameen Tahir
The following are details of my Areas of Effectiveness. The following are details of my Areas of Effectiveness English Language Proficiency, Organization Behavior , consumer Behavior and Marketing, Communication, Applied Statistics, Research Methods , Cognitive & Affective Processes, Cognitive & Affective Processes, Data Analysis in Research, Human Resources Management ,Research Project,
Social Psychology, Personality Psychology, Introduction to Applied Areas of Psychology,
Behavioral Neurosdence , Historical and Contemporary Issues in Psychology, Measurement in Psychology, experimental Psychology,
Business Ethics Business Ethics An introduction to business studies Organization & Management Legal Environment of Business Information Systems in Organizations Operations Management Global Business Policies Industrial Organization Business Strategy Information Management and Technology Company Structure and Organizational Management Accounting & Auditing Financial Accounting Managerial Accounting Accounting for strategy implementation Financial accounting Introduction to bookkeeping and accounting Marketing Marketing Management Professional Development Strategies Business Communications Business planning Commerce & Technology Human resource management General Management Conflict management Leadership Organizational Leadership Supply Chain Management Law Corporate Strategy Creative Writing Analytical Reading & Writing Other Expertise Risk Management Entrepreneurship Management science Organizational behavior Project management Financial Analysis, Research & Companies Valuation And any kind of Excel Queries.
4.70+
16+ Reviews
34+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Find the total capacitance CT of the network in figure. 3 F CT 4 uF 6 F 6 F 3 F
-
Using the pka values in Table 3.1, calculate the equilibrium constant for the following reaction. F- acting as a base toward the acid HCN
-
Using the pKa values in Table 17.1, predict the products of the following reactions: a. b. c. d. OCH3 CH3 NaOH CH3 Table 17.1 The pKa Values of the Conjugate Adds of the Leaving Groups of Carbonyl...
-
Complete the following table of basic calculations. For Percent Contribution Margin, use (P-MC)/P. Round to table standard. Price 18 17 16 15 14 13 j Quantity Demanded 600 a 800 900 1000 1100 1200...
-
Did the defendants conduct constitute ordinary negligence, meaning they are not liable under 519(b), or gross negligence, meaning they are liable?
-
Classic Ice Cream operates several stores in a major metropolitan city and its suburbs. Its budget and operating data for 2010 follow: Budgeted Data for 2010 Actual Operating Results in 2010 Flavor...
-
What is the purpose of practice aids in forensic and valuation services?
-
Sales of industrial vacuum cleaners at R. Lowenthal Supply Co. over the past 13 months are as follows: (a) Using a moving average with three periods, determine the demand for vacuum cleaners for next...
-
Problem 3 For a multiple linear regression model with 4 variables, where ~ Y = Bo+B1X1 + B2X2 + 3X3 + B4X4 + , N(0,2), what is the test statistic for each of the following tests? and what is the...
-
Write an equation for each of the following equilibria, and use Table 3.1 to identify the pK a value associated with the acidic species in each equilibrium. (a) Ammonia acting as a base toward the...
-
What is the dissociation constant of an acid that has each of the following pK a values? (a) 4 (b) 7.8 (c) 2
-
A bag contains five red chips, three white chips, and two blue chips. Three chips are to be selected at random, without replacement. Determine the probability that. The first two are red and the...
-
What action is taken by the Internal Revenue Service to punish individuals who engage in improper financial transactions involving nonprofits?
-
Ingrid Dodson is a staffing specialist at a K-12 tutoring company. The tutoring company is expanding beyond their current writing and math tutoring services and will also offer financial tutoring...
-
Find and share a resource that compares the Agile Manifesto to PMBOK standards and address the following (you will have to search outside of the reading assignments for this Module): Define Each What...
-
Adherence to and development of the Common Law is the cornerstone of the American legal system. Briefly explain how the common law developed over time, how the common law canon (a legal canon is a...
-
Element Corporation has 2 divisions: The Cushion Division and the Chair Division. You are the Vice President in charge of the Cushion Division. Your Cushion Division is a profit center. Each division...
-
In the accompanying graph, is the slope of curve AA positive or negative? Does the slope increase or decrease as we move along the curve from A to A? Answer the same two questions for curve BB. A' B '
-
How do individual companies respond to economic forces throughout the globe? One way to explore this is to see how well rates of return for stock of individual companies can be explained by stock...
-
When a mixture of methane and chlorine is irradiated, reaction commences immediately. When irradiation is stopped, the reaction gradually slows down but does not stop immediately. Explain.
-
Radical chlorination of pentane is a poor way to prepare 1-chioropentane, but radical chlorination of neopentane, (CH3)4C, is a good way to prepare neopentyl chloride, (CH3)3CCH2C1. Explain.
-
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination...
-
III. GregIFER inc., a Spain-based company, imports merchandise produced in Argentina. The price in the factory is 2,000,000. The expenses incurred during the transaction are: Transport costs from...
-
Pool Corporation ( ticker: POOL ) is the biggest swimming pool supplies distributor in U . S . Please go to its latest 1 0 - K filings and answer the following questions. What is POOL s % of...
-
You are a senior accountant in a tier 2 accounting firm based in Australia. One of the firm s clients, Avocado Ltd , has prepared a draft set of financial statements for the year ended 3 1 December 2...

Study smarter with the SolutionInn App


