What glycol undergoes oxidation to give each of the following sets of products? (a) HC HC C=0+0=
Question:
What glycol undergoes oxidation to give each of the following sets of products?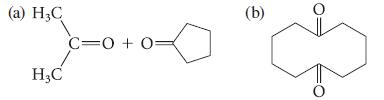
Transcribed Image Text:
(a) H₂C H₂C C=0+0= (b) O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
a C...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Each of the following pairs of compounds undergoes a Bronsted acid-base reaction for which the equilibrium lies to the right. Give the products of each reaction, and identify the acid, the base, the...
-
Each of the following pairs of compounds undergoes a Bronsted acidbase reaction for which the equilibrium lies to the right. Give the products of each reaction, and identify the acid, the base, the...
-
The current zero-coupon yield curve of semi-annually compounded rates for risk-free bonds is as follows: 1.0 Years 1.5 Years 2.0 Years 2.5 Years 3.0 Years 9.00% 10.20% 3.00% 4.00% 6.20% MATURITY 0.5...
-
On November 6 of Year 1, Havens Company provided services (on account) to a client located in Thailand. The contract price is 100,000 Thai baht. On November 6, the exchange rate was 50 baht for one...
-
Given the following information, calculate the change in cash for the year: Cash received from sale of company equipment.....$ 20,000 Cash paid for salaries................ 8,500 Cash paid for other...
-
Pick an industry and a product or service. Engage in a creative-thinking process, as outlined in Chapter 11, to generate an improved offering. Do the same to create an entirely new offering that uses...
-
A company is deciding which of two new thermostat systems to produce and sell. The Basic system has variable costs of $8.00 per unit, excluding sales commissions, and annual fixed costs of $520,000;...
-
Objectives: Your company is currently planning to automate many different system administration tasks. Your manager has asked you to research a system administration task and implement it using...
-
Using the abbreviations for lysine and SAM shown in Eq. 11.60a, write a curved-arrow mechanism for the formation of N,Ndimethyllysine. Assume that acids ( + BH) and bases (:B) are available as...
-
Give the product(s) expected when each of the following compounds is treated with periodic acid. OH OH | -CH-CH3 (b) | PhCHCHCHOH OH
-
In Exercises 2328, evaluate each factorial expression. 20! 2!18!
-
Estimate the binary diffusion coefficient for a ethanenitrogen mixture containing 60 mol% ethane at 310 K amd 60 atm absolute pressure. First, estimate the diffusivity at 1 atm. Then, use the...
-
A silica-based catalyst particle has a porosity p = 0.60. Estimate its thermal conductivity at 25C if the pores are (a) filled with air at 1 atm and (b) filled with liquid water.
-
An open tank, 3.0 m in diameter, contains liquid benzene at 37C exposed to air also at 37C and 1 atm absolute pressure. Assume that the rate of evaporation is controlled by diffusion through a...
-
It is March 13th and you just left the beach with your friends carrying along great memories of a fabulous Spring break. As your thoughts drift from sun-drenched beaches to mass transfer problems,...
-
A spherical naphthalene ball with an initial diameter of 1.2 cm slowly sublimes in a large volume of still air at 15C and 1 atm. At this temperature, the sublimation pressure of naphthalene is 0.036...
-
This chapter noted that returns on investment are taxable, whereas returns of investment are not taxable. What is the conceptual basis for this treatment? Cite examples of each type of return, and...
-
Design an experiment to demonstrate that RNA transcripts are synthesized in the nucleus of eukaryotes and are subsequently transported to the cytoplasm.
-
Lithocholic acid is an A-B cis steroid found in human bile. Draw Lithocholic acid showing chair conformations as in figure, and tell whether the hydroxyl group at C3 is axialequatorial. An A-B trans...
-
Compare the structures of lanosterol and cholesterol, and catalog the changes needed for the transformation.
-
The following model is that of cholic acid, a constituent of human bile. Locate the three hydroxyl groups, and identify each as axial or equatorial, is cholic acid an A t trans steroid or an A-B...
-
Do you think science is important on modern society? Explain your answer. Should we be concerned that so few enter STEM fields in the US ? Do some research on these topics and submit an essay for...
-
The Irving Corporation's stock trades currently at ( $ 20 ) per share with 10 million shares outstanding. Irving Corporation has four main business units. Units A & B are longstanding businesses that...
-
Your Company gives customers terms of 3 % off for payments within 1 0 days of invoicing and full price if paid in 4 0 days. So , on a $ 1 0 0 sale, a customer pays an extra $ 3 to pay 3 0 days later....

Study smarter with the SolutionInn App


