When a deuterium-substituted cyclohexene is subjected to CyP450-promoted oxidation, two different allylic alcohols are formed. Explain. D
Question:
When a deuterium-substituted cyclohexene is subjected to CyP450-promoted oxidation, two different allylic alcohols are formed. Explain.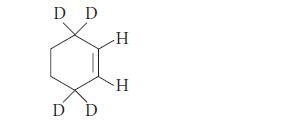
Transcribed Image Text:
D D D D H H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Remember that once the benzylic hydrogen or in this case deuterium ...View the full answer

Answered By

Nyron Beeput
I am an active educator and professional tutor with substantial experience in Biology and General Science. The past two years I have been tutoring online intensively with high school and college students. I have been teaching for four years and this experience has helped me to hone skills such as patience, dedication and flexibility. I work at the pace of my students and ensure that they understand.
My method of using real life examples that my students can relate to has helped them grasp concepts more readily. I also help students learn how to apply their knowledge and they appreciate that very much.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What is the effect of cultural commoditization and transformation on local traditions and customs? Give an example. Check Chapter 4 in the textbook for information on cultural commoditization and...
-
The Swern oxidation, shown in Fig. P10.61, is a very mild two-step procedure for the oxidation of primary and secondary alcohols. (a) How many electrons are involved in this oxidation? Explain. (b)...
-
Please help with the discusin questions ! I give thumbs up Case #1: Hailing a New Era: Haier in Japan As one of the most valuable brands in China, Haier designs,manufactures, and sells various home...
-
What would a profile look like across a restraining bend? Releasing bend?
-
Effects of gains and losses from sales of equipment on cash flows Exhibit 5.23 presents an abbreviated statement of cash flows for Largay Corporation for the current year (amounts in thousands)....
-
You have been asked to use the expected-value model to assess the risk in developing a new product. Each strategy requires a different sum of money to be invested and produces a different profit...
-
In Figure VI.1, what is the direction of the electric field in the region between the cathode and the target electrode? A. To the left B. To the right C. Toward the top of the page D. Toward the...
-
Find the future value of the following annuities. The first payment in these annuities is made at the end of Year 1; that is, they are ordinary annuities. (Note: See the hint to Problem 2-1. Also,...
-
This involves one person (or a small team) and can be completed in less than a day. One example is to use torque wrenches instead of adjustable wrenches. This statement BEST describes which of the...
-
(a) A compound A has the formula C 8 H 10 . After vigorous oxidation, it yields phthalic acid. What is the structure of A? (b) A compound B has the formula C 8 H 10 . After vigorous oxidation, it...
-
Explain how and why the product(s) would differ in the following reactions of trans-2-buten-1-ol. (1) Reaction with concentrated aqueous HBr (2) Conversion into the tosylate, then reaction with NaBr...
-
Calculate E' for the following reaction: Succinate + O Fumarate + HO 2
-
In 2004, Keith Jackson invested in a partnership known as Astonishing Discoveries, Ltd. He was a limited partner. Mr. Jackson paid $25,000 cash and, along with all other partners, signed a...
-
Thomas Settleton owned an interest as a general partner in LBO partnership. Settleton was not a material participant in the activity. His basis was $25,000 on the date he gifted the LBO partnership...
-
Janice Hoplin, MD, owned her own medical clinic. She also owned the office building in which the clinic was located. The medical activity generated $125,000 of net income. She managed the building,...
-
LCD Corporation purchased residential real property for $385,000 (exclusive of land cost) and placed it in service on August 16, 2019. Compute MACRS depreciation on the property for 2019 and 2020.
-
Define the term travel expenses as used for tax purposes. Define the term away from home as interpreted by the IRS.
-
Compute a one-way ANOVA on the following data. Determine the observed F value. Compare the observed F value with the critical table F value and decide whether to reject the null hypothesis. Use (...
-
Which of the following is NOT a magnetic dipole when viewed from far away? a) A permanent bar magnet. b) Several circular loops of wire closely stacked together with the same current running in each...
-
From the molecular masses and the relative intensities of their M and M + 1 peaks, suggest molecular formulas for the following compounds. M (m/z = 82; 37%), M + 1 (2.5%); contains C and H.
-
Suggest a structure for each of the ions corresponding to the following peaks in the EI mass spectrum of ethyl bromide, and give a mechanism for the formation of each ion. (The numbers in parentheses...
-
The physical basis of some carbon monoxide detectors is the infrared detection of the unique stretching vibration of carbon monoxide at 2143 cm-1. How many times per second does this stretching...
-
Fanciful Fan Company has five sales representatives and pays them a salary of $2,000 each per month. It also pays 15% commission on the sale price of each fan. Each fan is sold at a price of $100....
-
A partner sold a 25% interest in a partnership for $400,000 cash plus assumption of the partner's share of the partnership liabilities. The following additional information relates to the partnership...
-
Using the info provided, determine the statement balance. (Round your answer to 2 decimal places.) Balance last statement: Checks processed: Deposits received: Service charge: Statement balance:...

Study smarter with the SolutionInn App


