When the diethyl ester of a substituted malonic acid is treated with sodium ethoxide and urea, Veronal,
Question:
When the diethyl ester of a substituted malonic acid is treated with sodium ethoxide and urea, Veronal, a barbiturate, is formed (see Fig. P22.84). (Barbiturates are hypnotic drugs; some are actively used in modern anes-thesia.) Using the curved-arrow notation, give a mechanism for the Veronal synthesis.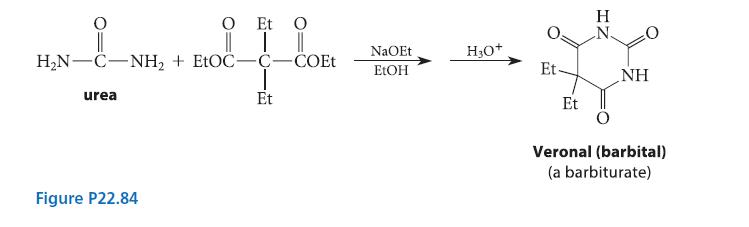
Transcribed Image Text:
O Et O _i_ || HN-C−NH, + EtOC—C−COEt Hi urea Et Figure P22.84 NaOEt EtOH H₂O+ Et Et IZ H NH Veronal (barbital) (a barbiturate)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
This reaction is essentially a type of crossedClaisen conden...View the full answer

Answered By

Shikha SharmaThakur
My teaching method is primarily focused on explaining practical scenarios which helps in quick and effective understanding.
I begin with basic concepts, followed by intensive practice in tutorials and examples.
I usually prefer quick tests to know students understanding of the subject.
I design my own examples in various stages of difficulty levels to help in students understanding in addition to the text book questions
Mathematics is to solve a task with a set of rules. I provide quick notes and rules to be refereed that can be helpful to solve questions of any difficulty level
I believe learning is better if you can visualize it, and hence in addition to providing real world examples, I prefer teaching using graphs and plots for enhanced understanding of the subject
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements: (a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism. (b)...
-
Give the mechanistic symbols (SN1, SN2, E1, E2) that are most consistent with each of the following statements: (a) Methyl halides react with sodium ethoxide in ethanol only by this mechanism. (b)...
-
When cis-1-bromo-4-tert-butylcyclohexane is treated with sodium ethoxide in ethanol, it reacts rapidly; the product is 4-tert-butylcyclohexene. Under the same conditions, trans-...
-
A newly issued 20-year maturity, zero-coupon bond is issued with a yield to maturity of 8% and face value $1,000. Find the imputed interest income in the first, second, and last year of the bonds...
-
Explain why the Merchandise Inventory account will usually require adjustment at year-end.
-
The balance sheet and income statement for Chico Electronics are reproduced below (tax rate is 40%). Required: Compute and interpret the following financial ratios of the company for Year 5: a....
-
Do analytical efforts, big data examinations, and textual analyses impact compliance and fraud deterrence? Provide an explanation for your opinion.
-
R. Santiago Co. uses special journals and a general journal. The following transactions occurred during May 2014. May 1 R. Santiago invested $40,000 cash in the business. 2 Sold merchandise to Lawrie...
-
What is the binary pattern represented by the following Differential Manchester encoding:
-
Using the curved-arrow notation, provide mechanisms for each of the reactions given in Fig. P22.87. H3C C-CH3 (c) (b) BrCHCO,CH3 I (CH)3 T BrCHCO,CH3 2 COEt KCO3 COEt NaOEt + 2 NaH HO CH3 DMF HO,...
-
A useful diketone, dimedone, can be prepared in high yield by the synthesis shown in Fig. P22.83. Provide structures for both the intermediate A (a Michael-addition product) and dimedone, and give a...
-
Compare the ES knowledge-refining system with knowledge improvement in machine learning.
-
It is a basic principle of tax law that income should be taxed to the individual who earns it. The income earner cannot transfer the income to someone else in order to escape paying tax on the...
-
Fourth Teaching: What does Krishna suggest about himself, about his true nature, in this teaching? (4.7- 10). How can one "do nothing at all" even when one acts? (4.20). What is the nature of...
-
How do you foresee a country for not paying taxes? Give and explain three (3) reasons why is it important for an employee to be knowledgeable of how his/her tax is computed.
-
You have been asked to produce a description of thepurposeandworkingof differential gearing for a small section of a children's book (target age 12-14) (This must be less than one hundred and fifty...
-
Using a current or former workplace as your example, answer the questions in the section of this chapter called Communication and Organizational Cultures about how to understand a workplace culture....
-
Prepare a table similar to Exhibit, which focuses on residual income. Use a 10 percent rate to compute the imputed interest charge. The table should show the residual income on the investment during...
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
Write structures for the major organic products from the following reactions. Show stereoisomers where applicable. (a) (b) (c) (d) C2 (1 equiv.) HBr (excess) (1) NaNH2 (2) PhCH2Br (1) 03 (2) HOAc
-
Show how 1-butyne could be synthesized from each of the following: (a) 1-Butene (b) 1-Chlorobutane (c) 1-Chloro-1-butene (d) 1, 1-Dichlorobutane (e) Ethyne and ethyl bromide
-
Starting with 2-methylpropene (isobutylene) and using any other needed reagents, outline a synthesis of each of the following: (a) (b) (c) (d) OR Br CI HO
-
This is the case of Cabot Oil & Gas Corporation v. Daugherty Petroleum, Inc. Question: please summarize and include the following; Fact: Judgment: Issue: Holding: General Analysis Applied Analysis:...
-
Assignment: Torts "We are living in unprecedented times" is a phrase that we became quite familiar with during the pandemic. One and a half years following the onset of Shelter-In-Place, we saw the...
-
The annual operation costs of an outdated environmental control device is $75,000. Under a proposed emission reduction plan, the installation of a new fan system will require an initial cost of...

Study smarter with the SolutionInn App


