Which of the following compounds, all known in nature, can be classified as terpenes? Show the isoprene
Question:
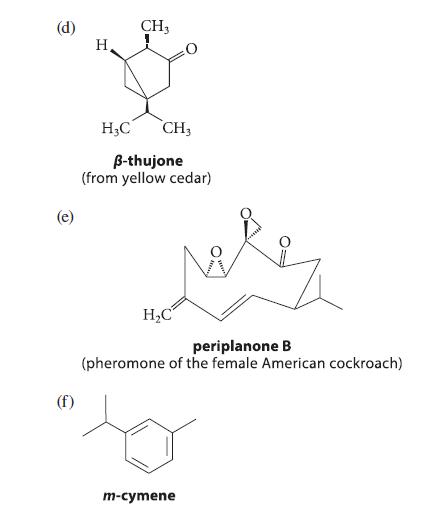
Which of the following compounds, all known in nature, can be classified as terpenes? Show the isoprene skeletons in each terpene.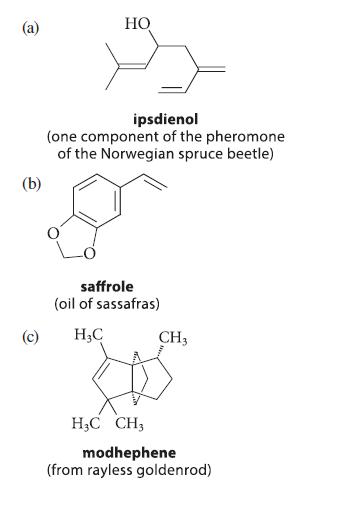
Transcribed Image Text:
(b) (c) HO ipsdienol (one component of the pheromone of the Norwegian spruce beetle) saffrole (oil of sassafras) H₂C CH3 H3C CH3 modhephene (from rayless goldenrod)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Compounds a d and e are terpenes The isoprene skeletons are shown ...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In the most general definition of an acid and a base, a Lewis acid is a compound that can accept an electron pair, and a Lewis base is a compound that can donate an electron pair. Which of the...
-
Show how each of the following compounds can be prepared from cyclohexene: a. b. c. d. OH
-
Which of the following compounds can be prepared by radical halogenation with little complication by formation of isomeric by-products? CI CI
-
Write a program that takes an integer command-line argument n and prints all the positive powers of 2 less than or equal to n. Make sure that your program works properly for all values of n.
-
Interpreting a statement of cash flows based on the direct method for presenting cash flow from operations Exhibit 5.32 shows the consolidated income statement s for CVS Caremark Corporation for...
-
How does a manager decide whether the work breakdown structure should be based on a tree diagram or the PERT diagram?
-
During the \(0.75 \mathrm{~s}\) that the tube is running, what is the electric power? A. \(7.0 \mathrm{~kW}\) B. \(700 \mathrm{~W}\) C. \(70 \mathrm{~W}\) D. \(7.0 \mathrm{~W}\) \(\mathrm{X}\) rays...
-
On July 1, 2014, Kellerman Company issued $5,000,000, 8%, 10-year bonds at $4,376,892. This price resulted in an effective-interest rate of 10% on the bonds. Kellerman uses the effective-interest...
-
Compute conversion costs given the following data: direct materials, $363,200; direct labor, $204,600; factory overhead, $192,100 and selling expenses, $39,100.
-
(a) Determine whether the following compound (zoapatanol, used as a fertility-regulating agent in Mexican folk medicine) is a terpene. (b) What product is obtained when zoapatanol is subjected to MnO...
-
Complete the stepwise process shown in Eq. 17.53 to give lanosterol, the product of Eq. 17.52. CH3 H3C H HO CH3 H CH3 H p! CH3 H3C T CH3 CH3 H3C H CH3 H3C- CH3 CH3 + CH3 H CH3 H3CC. CH3 H3C H...
-
What is the highest-order maximum for 400-nm light falling on double slits separated by 25.0 m?
-
Bradford is 12 years old and is eligible to be claimed as a dependent on his parents' return. In 2019, he received unearned income of $2,300. Bradford's itemized deductions totaled $150. Determine...
-
Allen has taxable income of $75,475 for 2019. Using the Tax Rate Schedules in the Appendix, compute Allen's income tax liability before tax credits and prepayments for each of the following filing...
-
Tom and Linda are married taxpayers who file a joint return. They have itemized deductions of $27,400 and four exemptions. Assuming an adjusted gross income of $40,000, what is their taxable income...
-
Research Problem. Sidney and Angelene were married in 2015. During 2019, they were both employed, each earning $132,000 for the year. They discovered that there was a significant tax savings if they...
-
The Tax Reform Act of 1986: a. Amended the Internal Revenue Code of 1954 b. Replaced the Internal Revenue Code of 1954 with the Internal Revenue Code of 1986 c. Replaced the Internal Revenue Code of...
-
A management consulting company presents a three-day seminar on project management to various clients. The seminar is basically the same each time it is given. However, sometimes it is presented to...
-
The comparative statements of financial position of Menachem NV at the beginning and end of the year 2019 appear below. Net income of ¬34,000 was reported, and dividends of ¬23,000 were paid...
-
Two of the compounds given in Fig. Pl 1.78 form epoxides readily when treated with -OH, one forms an epoxid slowly, and one does not form an epoxide at all. Identify the compound(s) in each category...
-
What stereochemical result would you expect if the (2s,3s)-stereoisomer of 3-bromo-2 butanol undergoes the same reaction?
-
Complete the following reaction by giving the major organic product dilute H SO CH CH CH OH (solvent)
-
In laparoscopy procedures gases is used for inflation abdominal wall is Nitrous oxide Oxygen Carbon Dioxide Helium
-
A biochemical marker of pre-eclampsia is:- Raised Hb level Raisedhematocrit level Raisedelectroyte level Raised uric acid level
-
Bayside Fishing Supply Co . is acquiring Fishing Lure Specialists for $ 2 5 , 2 5 0 in cash. Bayside Fishing Supply C 0 . has 2 , 3 5 0 shares outstanding at a market price per share of $ 3 9 ....

Study smarter with the SolutionInn App


