Which of the following compounds can be resolved into enantiomers at room temperature? Explain. (a) Z: (c)
Question:
Which of the following compounds can be resolved into enantiomers at room temperature? Explain.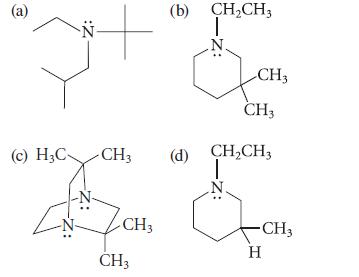
Transcribed Image Text:
(a) Z: (c) H₂C- CH3 CH3 CH3 (b) CH₂CH3 T CH3 CH3 (d) CH₂CH3 ☆ - CH3 H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
a The only stereocenter in the molecule is the nitrogen which rapidly u...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of the following compounds could be resolved into enantiomers at room temperature? Explain. (a) (b) CH2CH CH3)2 CH CH CH3 CH3
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,2,3,3, -tetramethylbutane
-
Which of the following compounds could in principle be resolved into enantiomers at very low temperatures? Explain. (a) Propane (b) 2,3-dimethylbutane (c) 2,2,3,3-tetramethylbutane
-
Freight and other handling charges on goods out on consignment are part of the cost of goods consigned. What is its appropriate account title in the income statement prepared by the consignor?...
-
Discuss trends impacting marketing and the implications of these trends on how marketers deliver value to customers.
-
Coyote Loco, Inc., a distributor of salsa, has the following historical collection pattern for its credit sales. 70 percent collected in the month of sale. 15 percent collected in the first month...
-
Use the September transaction data for Otis Carpenter, M.D., P.C. Requirements 1. Open the following T-accounts: Cash, Accounts receivable, Medical supplies, Accounts payable, Common stock, Service...
-
Mino Inc. manufactures chocolate syrup in three departments: Cooking, Mixing, and Bottling. Mino uses the weighted average method. The following are cost and production data for the cooking...
-
A.Identify a company offering this product and explain its orientation toward consumers . Does this company use undifferentiated, differentiated, or niche marketing? Explain B. Is this company...
-
Explain why 1-methylaziridine undergoes amine inversion much more slowly than 1-methylpyrrolidine. (What are the hybridization and bond angles at nitrogen in the transition state for inversion?)...
-
Draw a chair conformation for (S)-3-methylpiperidine showing the sp 3 orbital that contains the nitrogen unshared electron pair. How many chair conformations of this compound are in rapid...
-
Little Bite, Inc. manufactures dog biscuits for big dogs. Last month the company produced 18,000 biscuits. Using job order costing, determine the product unit cost for one dog biscuit based on the...
-
Word Problem 1-54 (Static) [LU 1-2 (1)] As of mid-September 2021, 229,552,716 worldwide cases of coronavirus were reported by www.worldometers.info. It was also reported 206,248,522 have recovered...
-
The answer should demonstrate your ability to analyze the facts in the question, to tell the difference between material facts and immaterial facts, and to discern the points of law and facts upon...
-
The altitude of a satellite in an elliptical orbit around theearth is 1600 km at apogee and 600 km at perigee. A. Determine the eccentricity of the orbit. B. Determine the orbital speeds at perigee...
-
For each of the following production functions, determine whether it exhibits increasing, constant or decreasing returns to scale: a) Q = 2K+L b) Q=3L+L/K c) Q = Min(2K,L) d) Q=L*K
-
2. The upper and lower limbs of the human body have the exact same body plan. Answer the questions below to diagram out that body plan of human limbs. a. How many bones are in the upper portion of...
-
Identify some of the differences between SFP presentations around the world.
-
Suppose that a flow network G = (V, E) violates the assumption that the network contains a path s t for all vertices V. Let u be a vertex for which there is no path s u t. Show that there must...
-
Show the products, including stereo chemistry, of these SN1 reactions: a) CHCH Ph CH3 + CHOH HC b) CHCH H CH, -CH-C-CI+ CHCOH CH
-
Explain whether this reaction would follow the SN1 or the SN2 mechanism and then explain which reaction is faster: CI a) CHCH + OH HO CHOH CH3 b) CHC-Br + CHCO CH, O-SOCH3 or CHCH CH CO,H or + OH CH3...
-
(a) Show all the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of electrons in each step of the mechanism. (b) Show a free energy versus reaction...
-
Sheridan Company Ltd. publishes a monthly sports magazine, Fishing Preview. Subscriptions to the magazine cost $28 per year. During November 2025, Sheridan sells 7,200 subscriptions for cash,...
-
At April 30, Barker Co.'s cash balance was $10,053 before adjustments, and its April 30 bank statement balance was $9,823. Check number 205 was issued April 10 in the amount of $36 for supplies, but...
-
Direct labor $134,730 Indirect labor $19,860 Utilities $14,570 Supplies $4,980 Equipment depreciation $54,080 Factory rent $8,700 Property taxes $2,100 Factory administration $26,470 Packaging...

Study smarter with the SolutionInn App


