Which of the following four structures represent constitutional isomers of the same molecule, and which one is
Question:
Which of the following four structures represent constitutional isomers of the same molecule, and which one is neither isomeric nor identical to the others? Explain your answers.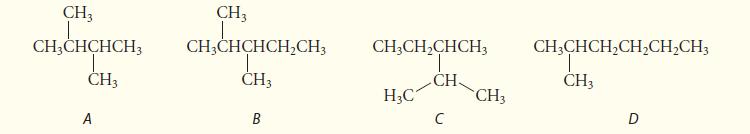
Transcribed Image Text:
CH3 CH3CHCHCH3 CH3 A CH3 CH3CHCHCH₂CH3 CH3 B CH3CH₂CHCH3 T CH H₂C C CH3 CH3CHCH₂CH₂CH₂CH3 CH3 D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
Compounds must have the same molecular formula to be either identical or isomeric Structure A has a ...View the full answer

Answered By

Raunak Agarwal
Teaching is my hobby and now my profession. I teach students of CA and CFA(USA) in batches of 100 students and have a 5 year experience.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What did you learn about Essential Business Concepts?
-
Which of the following structures represent the same compound? Which ones represent different compounds? (a) (b) (c) (d) (e) (f) (g) Name the structures given in Problem 3-33, parts (a), (c), (e),...
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
Suppose that Canada produces two goods: lumber and fish. It has 18 million workers, each of whom can cut 10 feet of lumber or catch 20 fish each day. a. What is the maximum amount of lumber Canada...
-
Researchers can draw various conclusions from a set of data. How do you know how to shape conclusions and recommendations?
-
Your company has $200,000 to invest and has identified the following three investments. Investment A requires an initial investment of $130,000 and has an annual rate of return of 12%. Investment B...
-
Use the technique discussed in Section9.4 to develop a GEE approach for zeroinflated Poisson model for count responses in longitudinal studies. Section9.4: 9.4 Marginal Models for Longitudinal Data...
-
Eleanor's Computers is a retailer of computer products. Using the financial data provided, complete the financial ratio calculations for 2010. Advise management of any ratios that indicate potential...
-
John just sold the home where he's lived for eight years and made a nice profit. Which of these statements about the profit he made is FALSE? Unset starred question He can include depreciation costs...
-
(a) Draw a Newman projection for each staggered and eclipsed conformation about the C2C3 bond of isopentane, a compound containing a branched carbon chain. Show all staggered and eclipsed...
-
Suppose you take a trip to a distant universe and find that the periodic table there is derived from an arrangement of quantum numbers different from the one on Earth. The rules in that universe are...
-
Use the data in file agpop.dat for this exercise. Let yi be the value of acres92 for unit i and xi be the value of acres87 for unit i. Draw an SRS of size 400. Now generate missing data from your...
-
Tim Trepid is highly risk-averse while Mike Macho actually enjoys taking a risk. Investments Returns: Expected Value Standard Deviation Buy stocks $ Buy bonds Buy commodity futures Buy options 9,700...
-
1. Why do you think Everett set such an ambitious goal for transforming its fleet? Based on its achievements so far, do you think it is likely to meet that goal? Why or why not? 2. What additional...
-
identify three to five of the most important turning points in your career that have led to where you are now- serendipitous moments or opportunities that turned your career in another direction....
-
1. Write about a experiences working as a leader and/or with a leader: try to describe your experience according to trait, situational and functional perspectives. How might these different ways of...
-
Identify and give examples to illustrate the following aspects of customer relationship management, enterprise resource management, and supply chain management systems: a. Business processes...
-
How does the problem of limited and bundled choice in the public sector relate to economic efficiency? Why are public bureaucracies alleged to be less efficient than private enterprises?
-
Shreemaya Hotel in !adore was facing a problem of low demand for its rooms due to off season. The Managing Director (MD) of the hotel, Mrs. Sakina was very worried. She called upon the Marketing...
-
Show all the steps in the mechanism for this reaction. What substitution product(s) would also be formed in thisreaction? Br E:OH
-
Show the products of this reaction. How would the composition of the products change if t-BuO ? in t-BuOH were used in place of ethoxide ion in ethanol? ELOH + CH,CH,0
-
All of the stereo isomers of 1, 2, 3, 4, 5, 6-hexachlorocyclohexane have very similar rates of E2 reaction except the following stereo isomer, which reacts about 7000 times more slowly than the...
-
Identify any proposed or suggested changes in service financing and how the changes could reflect the concept of fiscal equivalence?
-
The cost of the "Ice Power" system is $8000 and the government will pay 30% of the cost of the system. The company guarantees the system's operation for 15 years and the salesman says the system will...
-
Husband Wife Total Properties acquired before the marriage Properties for exclusive use 70,000.00 20,000.00 90,000.00 Other properties acquired 530,000.00 380,000.00 910,000.00 Properties acquired...

Study smarter with the SolutionInn App


