Which of the two compounds in each of the following sets should react more rapidly in a
Question:
Which of the two compounds in each of the following sets should react more rapidly in a nucleophilic aromatic substitution reaction with CH3O¯ in CH3OH? Explain your answers.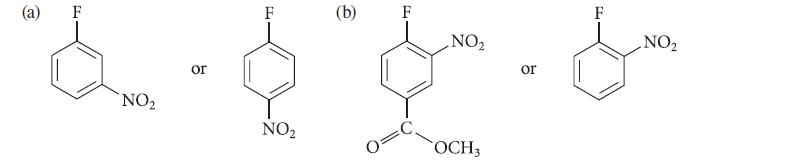
Transcribed Image Text:
(a) NO₂ or F NO₂ (b) NO₂ OCH3 or PL F NO₂
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a The second compound pfluoronitrobenzene reacts mo...View the full answer

Answered By

Nishtha Goel
have more than 6 years of teaching experience in ICSE and CBSE School of Subject Maths, Science and English. Also I have done MSc Chemistry and MA English and a little experience of process Control and Instrumentation.
For a question to solve, you first have a basic knowlegde and Understanding of the subject which I will provide to you. Moreover to understand the topic discussion plays an important role, when we discuss a certain topic with someone many questions arise in our mind and to solve that questions, I am available for you.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Explain how you could distinguish between the two compounds in each of the following sets using only 13C NMR spectroscopy. Trans - 1,2-cy clohexanediami ne and trans - 1,4-cyclohexanediamine
-
Which of the two isomers in each of the following sets should have the greater basicity at the carbonyl oxygen? Explain. H,C CHCHC- OCH or HCC-o-CH CH CH
-
(a) The following resonance-stabilized ion can protonate to give two different constitutional isomers. Give their structures and give the curved-arrow notation for their formation. (b) One of the...
-
Consider a situation with two countries that have abatement cost functions : for j=L and j=H. The countries have identical damage functions D(E)= d.E.For each country the parameters s j are drawn...
-
Jay has been posted to a remote region of space to monitor traffic. Toward the end of a quiet shift, a spacecraft goes by, and he measures its length using a laser device, which reports a length of...
-
The Murphy Company prepared a trial balance on the following partially completed worksheet for the year ended December 31, 2007: Additional information: (a) The equipment is being depreciated on a...
-
Measure the effect of cache conflicts on real-time execution time. First, set up your system to measure the execution time of your real-time process. Next, add a background process to the system. One...
-
At the end of January, Mineral Labs had an inventory of 775 units, which cost $12 per unit to produce. During February, the company produced 900 units at a cost of $16 per unit. If the firm sold...
-
Complete in Matlab, MATLAB, MATLAB % Given data T = 350; % Temperature in Kelvin P = 9.4573; % Pressure in bar Tc = 425.1; % Critical temperature in Kelvin Pc = 37.96; % Critical pressure in bar R =...
-
Calculate the oxidation state of the metal in each of the following complexes. (a) O Mn-O- permanganate (b) Pd(PPH3)4 tetrakis(triphenylphosphine) palladium (c) CpFe ferrocene
-
Noting the LX character of the allyl ligand in Table 18.1, sketch the allylmetal interaction, showing both L-type and X-type bonds. Use M as a general metal. TABLE 18.1 Some Typical Ligands Used in...
-
McGuire Corporation began operations in 2021. The company purchases computer equipment from manufacturers and then sells to retail stores. During 2021, the bookkeeper used a check register to record...
-
Marianos Hacienda Ranch is a Mexican restaurant in Dallas. Founder Mariano Martinez is not only known for serving up great Mexican food, but also for being the inventor of the worlds first frozen...
-
The five busiest airports in the United States are in Atlanta, Chicago, Los Angeles, Dallas-Ft. Worth, and New York City. At these airports there are more than 450 food outlets, with each airport...
-
Wealth distribution is not only calculated for households throughout the United States, but also by state. According to a 2010 study by Insidermonkey.com, Washington, D.C. ranked highest among all 50...
-
Mona Stangley owns an organic egg farm just outside of Gilbert, Texas. At present, she pays her workers an hourly wage to inspect and package her organic eggs in egg cartons. She is considering...
-
Included in the Tax Cuts and Jobs Act of 2017 was the elimination of the individual mandate penalty for not having health insurance. Starting in 2019, individuals who do not have health insurance...
-
A western city has 18 police officers eligible for promotion. Eleven of the 18 are Hispanic. Suppose only five of the police officers are chosen for promotion and that one is Hispanic. If the...
-
Smthe Co. makes furniture. The following data are taken from its production plans for the year. Required: 1. Determine the hazardous waste disposal cost per unit for chairs and for tables if costs...
-
For each reaction, estimate whether ÎSo for the reaction is positive, negative, or impossible to predict. a. (b) The formation of diacetone alcohol: c. heat C10H22--> n-decane C3H6 + C7H16...
-
(a) Propose a mechanism for the free-radical chlorination of ethane, (b) Calculate ÎHo for each step in this reaction. (c) Calculate the overall value of ÎHo for this reaction. hu CH3...
-
Draw Lewis structures for the following free radicals. (a) The ethyl radical, (b) The tert-butyl radical, (CH3)3C (c) The isopropyl radical (2-propyl radical) (d) The iodine atom CH3 CH2
-
For the following LCL shipments calculate the chargeable weight/measure the freight charges for each shipment including applicable surcharges (CAF is % of O/F charges, BAF & OHC are charged on w/m,...
-
Can you provide a significant historical event in the evolution of Supply Chain Management, and identify a key driver in the evolution of Supply Chain Management form (1900 - 1910 ) or (1935 to 1945)...
-
Explain the video and reading detail. Also Read the case provided below and answer questions Video: Supply Chain Risk Management. Supply Chain Risk Management (4:38 mins) Squire, B. (2013,...

Study smarter with the SolutionInn App


