Within each series, arrange the compounds according to increasing rates of their reactions by the S N
Question:
Within each series, arrange the compounds according to increasing rates of their reactions by the SN1–E1 mechanism. Explain your reasoning.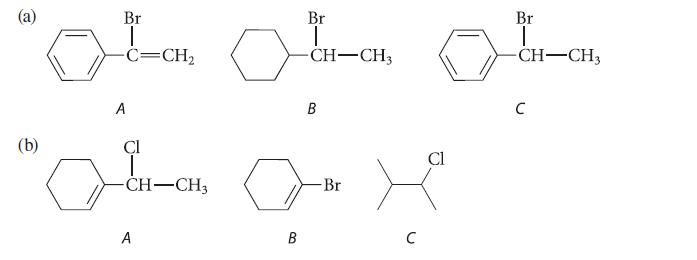
Transcribed Image Text:
(b) Br -C=CH, A Cl امي مسلم -CH-CH3 A Br | -CH-CH3 B B Br لى C Cl Br -CH-CH3 C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a b The o...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Within series, arrange the compounds according to increasing rates of their reactions by the SN1 - E1 mechanism. Explain your reasoning. CH CH Br
-
Within each series arrange the compounds in order of increasing stability: C(CH3)3 HO CH,CH(CH)
-
Within set, rank the compounds in order of increasing rates of their SN2 reactions. Explain your reasoning. 1-bromocyclohexene, bromocyclohexane, 1-(bromomethyl)cyclohexene
-
The example images in the text for Fade do not quite line up in the vertical direction (the mandrill's mouth is much lower than Darwin's). Modify Fade to add a transformation in the vertical...
-
The proper mean lifetime of a muon is 2 s. Muons in a beam are traveling at 0.999c. (a) What is their mean lifetime as measured in the laboratory? (b) How far do they travel, on average, before they...
-
What is an audit and why is the auditors report an important item of information?
-
Using a CPU that runs an operating system that uses EDF, try to get the CPU utilization as close to 100% as possible without failing. Try a variety of data arrival times to determine how sensitive...
-
The management of AAAA Company has observed that the companys cash outflows have been increasing much more rapidly than its inflows. Management cannot understand the change; from its perspective, it...
-
What are the mechanisms underlying immuno senescence and how does age-related decline in immune function impact susceptibility to infections and vaccine responsiveness?
-
Noting the LX character of the allyl ligand in Table 18.1, sketch the allylmetal interaction, showing both L-type and X-type bonds. Use M as a general metal. TABLE 18.1 Some Typical Ligands Used in...
-
Within each set, rank the compounds in order of increasing rates of their S N 2 reactions. Explain your reasoning. (a) Benzyl bromide, (3bromopropyl) benzene, pbromotoluene (b) 1bromocyclohexene,...
-
Three forces with magnitudes of 70pounds, 40 pounds, and 60 pounds act on an object at angles of 30, 45, and 135, respectively, with the positive x-axis. Find the direction and magnitude of the...
-
Most casino game dealers in Las Vegas are paid minimum wage, and therefore rely on tips for a large portion of their income. Within the past few years, a majority of Las Vegas casinos have instituted...
-
If in fact the Chinese government was allocating the rights to export under a quota to the most productive firms, what would you expect to see happen once the quota is lifted?
-
A citizens group in the Pacific Northwest has the following statement in its charter: Our goal is to ensure that large, powerful corporations pay their fair share of taxes in this country. To...
-
New PhDs in economics entering the job market find that academic jobs (jobs teaching at colleges and universities) pay about 30 percent less than nonacademic jobs such as working at a bank or a...
-
Suppose that in 2019, Congress passed and the president signed a new simple income tax with a flat rate of 20 percent on all income greater than $35,000 (no tax on the first $35,000). Assume that the...
-
Shown here are the top 10 U.S. cities ranked by number of rooms sold in a recent year. Suppose four of these cities are selected randomly. a. What is the probability that exactly two cities are in...
-
A parking lot charges $3 for the first hour (or part of an hour) and $2 for each succeeding hour (or part), up to a daily maximum of $10. (a) Sketch a graph of the cost of parking at this lot as a...
-
When a small piece of platinum is added to a mixture of ethene and hydrogen, the following reaction occurs: Doubling the concentration of hydrogen has no effect on the reaction rate. Doubling the...
-
(a) Draw the reaction-energy diagram for the reverse reaction: CH3 + HCl CH4 + Cl (b) What is the activation energy for this reverse reaction? (c) What is the heat of reaction (H) for this reverse...
-
Draw a reaction-energy diagram for the following reaction: CH3 + Cl2 CH3Cl + Cl The activation energy is 4 kJ mol (1 kcal mol), and the overall Ho for the reaction is -109kJ/mole (-26 kcal/mol) (b)...
-
For the following LCL shipments calculate the chargeable weight/measure the freight charges for each shipment including applicable surcharges (CAF is % of O/F charges, BAF & OHC are charged on w/m,...
-
Explain the video and reading detail. Also Read the case provided below and answer questions Video: Supply Chain Risk Management. Supply Chain Risk Management (4:38 mins) Squire, B. (2013,...
-
Question: Design a SWOT and TOWS analysis for Dutch Brothers Coffee Resources What is a SWOT Analysis Learn with Shopify. https://youtu.be/0K_MjuT2Cb0 Laksham K. Unmanned aerial vehicle (drones) in...

Study smarter with the SolutionInn App


