Work Study Problem 3.5 for the reverse of each reaction 3.16ac. CH3 H3C-C Br: I CH3 A
Question:
Work Study Problem 3.5 for the reverse of each reaction 3.16a–c.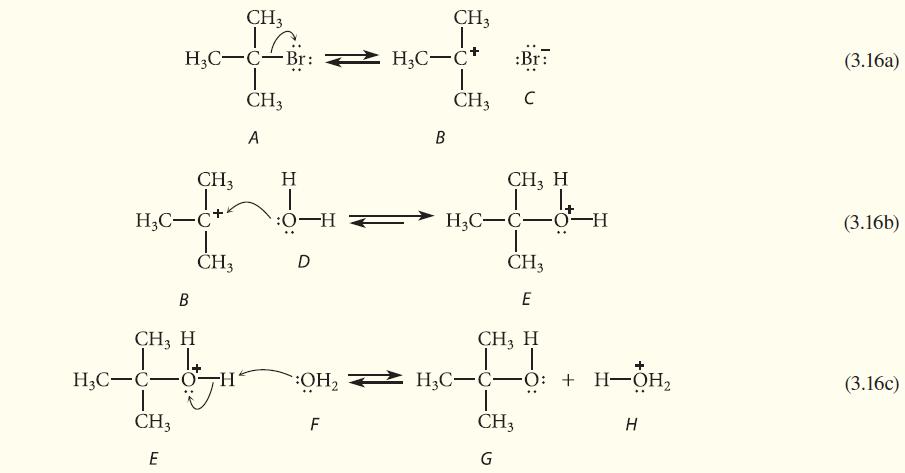
Transcribed Image Text:
CH3 H3C-C Br: I CH3 A CH3 H₂C-C+ B CH, H CH3 H3C-C-O -H I CH3 E H :0-H D F CH3 H₂C-C+ B T CH3 :Br C CH, H H3C-C-0-H CH3 E CH, H I OH₂ H3C-C-O: + H3C-CO: + H-OH₂ | CH3 G H (3.16a) (3.16b) (3.16c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a The reverse of reaction 316a is a Lewis acidbase association reaction ther...View the full answer

Answered By

Muhammad adeel
I am a professional Process/Mechanical engineer having a vast 7 years experience in process industry as well as in academic studies as a instructor. Also equipped with Nebosh IGC and lead auditor (certified).
Having worked at top notch engineering firms, i possess abilities such as designing process equipment, maintaining data sheets, working on projects, technical biddings, designing PFD and PID's etc.
Having worked as an instructor in different engineering institutes and have been involved in different engineering resrearch projects such as refinery equipment designing, thermodynamics, fluid dynamics, chemistry, rotary equipment etc
I can assure a good job within your budget and time deadline
4.90+
52+ Reviews
60+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-4. Ivan and Irene paid the following in 2012 (all by check or can otherwise be...
-
An autoglass company located in Detroit produces mirrors for automobiles. The company supplies auto mirrors to a nearby Ford (The Ford Motor Company, Dearborn, MI) plant. The demand for mirrors from...
-
Assume that the Tarasoff case is precedent. (In fact, many states have followed the ruling.) Make an argument that Ingram is liable.
-
Does being off by 150% in the estimate for human resources required for a project surprise you? What do you think happened? How do you think they managed to accommodate this change without exceeding...
-
The accountant for Metal Main, Inc., posted adjusting entries (a) through (e) to the accounts at August 31, 2010. Selected balance sheet accounts and all the rev enues and expenses of the entity...
-
An investor bought common stock of Microsoft Corporation on three occasions at the following prices. Calculate the average price per share at which the investor bought these shares. Date January 2009...
-
Lozano Chip Company: Balance Sheet as of December 31, 2016 (Thousands of Dollars) Cash $225,000 Accounts payable $601,866 Receivables 1,575,000 Notes payable 326,634 Inventories 1,125,000 Other...
-
What is the pK a of an acid that has each of the following dissociation constants? (a) 10-3 (b) 5.8 10-6 x (c) 50
-
Write a Brnsted acidbase reaction in which act as conjugate acidbase pairs. H/-:H and CH,H/CH,:-
-
Why is an understanding of management important even if you dont plan to be a manager?
-
BCC iron has a lattice parameter of 0.2866 nm. Carbon with a radius of 0.071 nm fills all the tetrahedral sites of the iron crystal. What is the atomic percentage of carbon?
-
You are provided with the following information relating to the Consolidated Fund for the year ended 3 1 ^ st December,Additional Information 1 . The following has been extracted from the 2 0 1 9...
-
A man stipulates in his will that $50,000 from his estate is to be placed in a fund from which his three daughters are each to receive the same amount when aged 21. When the man dies the girls are...
-
1. Explain the pre-conditions under which collective bargaining can be practiced in Jamaica. Cite laws, regulations and conventions to support your answer. [10 marks] 2. The contract of employment is...
-
The total debt ratio is currently 69.57%. If the net income and total assets stay the same, but new shares are issued to pay off the debt, and the total debt ratio decreases to 50%, what would the...
-
Consider the following data and corresponding weights. xi Weight (wi) 3.2 ......... 6 2.0 ......... 3 2.5 ......... 2 5.0 ......... 8 a. Compute the weighted mean. b. Compute the sample mean of the...
-
XYZ Inc. a calendar year, accrual basis corporation, had the following items during 2021: Gross revenue from operations Cost of goods sold $420,000 ($180,000) $9,000 LT capital gain .LT capital...
-
The reaction of hydroxide ion with chloromethane to yield methanol and chloride ion is an example of a general reaction type called a nucleophilic sub- Situation reaction: HO- + CH3C1 CH3OH + C1-...
-
Ammonia reacts with acetyl chloride (CH3COCI) to give acetamide (CH3CONH2). Identify the bonds broken and formed in each step of the reaction, and draw curved arrows to represent the flow of...
-
The naturally occurring molecule ?-terpineol is biosynthesized by a route that includes the following step: (a) Propose a likely structure for the isomeric carbocation intermediate. (b) Show the...
-
Determine how much of the original amount borrowed remains unpaid. Subtract the amount of the principal that has been repaid (from step 3) from the original amount borrowed: (Note: Round your answer...
-
Create your own life map (it could be a drawing, bullet points, paragraph or time line). Include things like important events, influential people, challenges and successes, and any other learning...
-
Cho Chen from Dallas, Texas, has borrowed $1,500 for 12 months plus an additional finance charge of $240. She is scheduled to pay equal monthly installments of $145 ($1,740 / 12). Assume that Cho...

Study smarter with the SolutionInn App


