A compound with molecular formula C 11 H 14 O 2 exhibits the following spectra ( 1
Question:
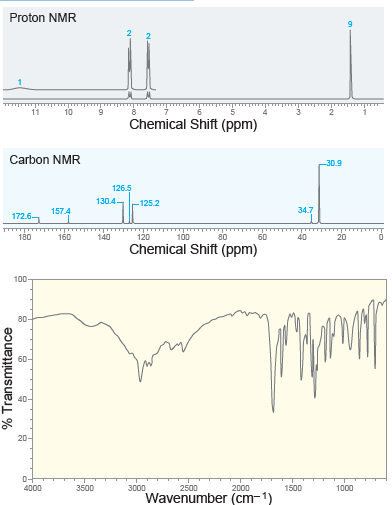
Transcribed Image Text:
Proton NMR 11 Chemical Shift (ppm) Carbon NMR 30.9 126.5 130.4- -125.2 34.7 157.4 172.6 120 100 80 180 160 140 60 40 Chemical Shift (ppm) 100 80- По 60- 40- 20- 0- 4000 3500 3000 2500 2000 1500 1000 Wavenumber (cm-1) % Transmittance
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)

Answered By

Gauri Hendre
I worked as EI educator for Eduphy India YT channel. I gave online tutorials to the students who were living in the villages and wanted to study much more and were preparing for NEET, TET. I gave tutions for topics in Biotechnology. I am currently working as a tutor on course hero for the biochemistry, microbiology, biology, cell biology, genetics subjects. I worked as a project intern in BAIF where did analysis on diseases mainly genetic disorders in the bovine. I worked as a trainee in serum institute of India and Vasantdada sugar institute. I am working as a writer on Quora partner program from 2019. I writing on the topics on social health issues including current COVID-19 pandemic, different concepts in science discipline. I learned foreign languages such as german and french upto A1 level. I attended different conferences in the science discipline and did trainings in cognitive skills and personality development skills from Lila Poonawalla foundation. I have been the member of Lila poonawalla foundation since 2017. Even I acquired the skills like Excel spreadsheet, MS Office, MS Powerpoint and Data entry.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
When a compound with molecular formula C11H14O2 undergoes acid-catalyzed hydrolysis, one of the products that is isolated gives the following 1H NMR spectrum. Identify the compound. 10 9 4
-
A compound with molecular formula C4H6O gives the infrared spectrum shown in Figure 13.34. Identify the compound. 3.5 4 5 12 13
-
Identify the following compounds. (Relative integrals are given from left to right across the spectrum.) a. The 1H NMR spectrum of a compound with molecular formula C4H10O2 has two singlets with an...
-
The passage indicates that the late 1850s Democrats: F. Were all Southern slaveholders who wanted to expand slavery into the territories. G. Used legislation in the early 1850s to support their...
-
What is breach of contract? What remedies are available to a party injured by a breach?
-
In deciding to add or drop customers or to add or discontinue branch offices or business divisions, what should managers focus on and how should they take into account allocated overhead costs?
-
Explain the primary activities of the (a) timekeeping and (b) payroll departments.
-
City Sights, Ltd., operates a tour and sightseeing business. Its trademark is the use of trolley buses. Each vehicle has its own identity and is specially made for the company. Gridlock, the oldest...
-
Computing the Predetermined Overhead Rate [LO3 - CC5] Harris Fabrics computes its predetermined overhead rate annually on the basis of direct labour-hours. At the beginning of the year, it estimated...
-
Giant pharmaceutical manufacturers such as Merck & Company and Pfizer have traditionally relied heavily on the doctor-to-patient channel to promote their products. In essence, this channel structure...
-
An ideal gas in a piston and cylinder assembly with adiabatic walls undergoes an expansion against a constant external pressure. Are S, S surroundings , and S total positive, negative, or zero?...
-
Is the equation valid for an ideal gas? Tf PV; -V;) T; Cy dT Lav = C, n2v, -v) %3D AS =
-
On January 1, 2016, Van Company had a balance of $800,000 in its Bonds Payable account. During 2016, Van issued bonds with a $300,000 face value. There was no premium or discount associated with the...
-
What is the price you would have to pay for a $1,000 bond that you purchased with a noncompetitive bid at an auction for 30-year Treasury Bonds where the stop-out yield was 4.120%? Please answer in...
-
What are the three market perception ratios and results for the state closing between 2021-2023 for Apple and Microsoft? It must include the calculations, formulas and analysis of said perception for...
-
There is a Treasury bill with a face value of $1million that matures in 45 days. Its quoted price is .012. A. What is its price in dollars and cents (remember that the face value is $1million)? B....
-
1. How much will you have saved after 6 years by contributing $1,200 at the end of each year if you expect to earn 11% on the investment? Insert your answer. 2. A business owner plans to deposit...
-
If one of the advantages of NPV is that it helps the management of the company in making better decisions by considering the time value of money, then considering the time value of money, is it...
-
Calculate the value of the multiple integral. where D is the region in the first quadrant bounded by the parabolas x = y 2 and x = 8 - y 2 dA D
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
What is the expected substitution product (including its stereochemical configuration) in the SN2 reaction of potassium iodide in acetone solvent with the following compound? (D = 2H = deuterium, an...
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
(a) Give the structure of the S*2 reaction product between ethyl iodide and potassium acetate. H,C-C potassium acetate
-
Dollar-Value LIFO On January 1, 2018, Sato Company adopted the dollar-value LIFO method of inventory costing. Sato's ending inventory records appear as follows: Year Current Cost Index 2018 $31,600...
-
How many standards are included in the ELP accreditation standards? What stands out to you about the different areas? What are the topic areas under administration and leadership? What are the...
-
What is performance management systems? Do the companies where you (or others you know) have worked used performance management systems rather than performance appraisal systems? If yes, what kind of...

Study smarter with the SolutionInn App


