All of the following acid-base reactions are reactions that we will study in greater detail in the
Question:
(a)
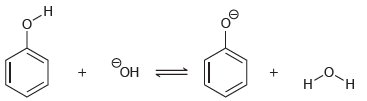
(b)
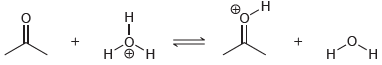
(c)
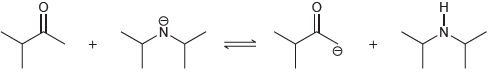
(d)
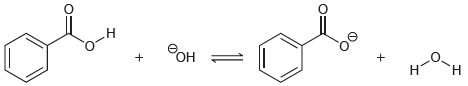
Transcribed Image Text:
Н eон Н H. H.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
a b c d Aci...View the full answer

Answered By

Antony Sang
I am a research and academic writer whose work is outstanding. I always have my customer's interests at heart. Time is an important factor in our day to day life so I am always time conscious. Plagiarism has never been my thing whatsoever. I give best Research Papers, Computer science and IT papers, Lab reports, Law, programming, Term papers, English and literature, History, Math, Accounting, Business Studies, Finance, Economics, Business Management, Chemistry, Biology, Physics, Anthropology, Sociology, Psychology, Nutrition, Creative Writing, Health Care, Nursing, and Articles.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 ? (b) CH3OH + NaNH2 ? (c) CH3NH3 + C1- +NAOH ?
-
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the equation:
-
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the equation:
-
McGuire Industries prepares budgets to help manage the company. McGuire is budgeting for the fiscal year ended January 31, 2021. During the preceding year ended January 31, 2020, sales totaled $9,200...
-
Obtain an annual report from a publicly traded corporation that is interesting to you. Be sure the company has property and equipment, intangible assets , and long-term debt on its balance sheet....
-
Determine dryness fraction of steam supplied to a separating and throttling calorimeter. Water separated in separating calorimeter \(=0.2 \mathrm{~kg}\) Steam discharged from throttling calorimeter...
-
For each of the following questions appearing on an internal accounting control questionnaire, assume that a "No" answer is given: 1. Is there separation of duties between the journalizing of...
-
Develop a communication plan for an airport security project. The project entails installing the hardware and software system that (1) Scans a passengers eyes. (2) Fingerprints the passenger. (3)...
-
What are the the differences between a Micro-manager and an Investor. Provide examples of each.
-
At the end of June, Rob Falco decided to open his own computer service. Analyze the following transactions he completed by recording their effects in the expanded accounting equation. a. Invested...
-
Identify any formal charges in the following structures: a. b. c. d. -N=N:
-
Each of the following mechanisms contains one or more errorsthat is, the curved arrows may or may not be correct. In each case, identify the errors and then describe what modification would be...
-
Identify each of the properties illustrated in Problems 2732. a. 5 + 7 = 7 + 5 b. 5 1 / 5 = 1
-
What might be some additional thoughts concerning planning and diversification of assets to lessen market volatility?
-
Examine the following yield curve from Monday. What do we expect the two-year rate to be five years from now? Assume zero-coupons. 1yr 4.03 2yr 3yr 5yr 7yr 10yr 20yr 30yr 3.96 3.94 3.75 3.69 3.57...
-
Suppose that Memorial Hospital has four projects. A summary of the projects is as follows: Project Amount Invested Market Beta Walk-in Clinic $1,500,000 1.3 MRI Facility $2,500,000 .7 Clinical...
-
The option adjusted spread (OAS) of a callable bond is zero. Is it true that this callable's yield equals that of the noncallable counterpart? Explain your answer.
-
The 0.5-year and 1-year risk free spot interest rates are 5% and 6% per year. Assume semi-annual compounding. Compute the 0.5- and 1-year zero coupon bond prices that pays $1 face value at maturity.
-
Solve the system using the inverse of the coefficient matrix. -x + y = 1 2x - y = 1
-
What is the mode?
-
The 1H NMR peak for the hydroxyl proton of alcohols can be found anywhere from d 0.5 to d 5.4. Explain this variability.
-
(a) Show that replacing each of the CH2 protons by some group Q in the (S) enantio-mer of 2-butanol leads to a pair of diastereomers, as it does for the (R) enantiomer. (b) How many chemically...
-
The 1H NMR study of DMF (N, N-dimethylformamide) results in different spectra according to the temperature of the sample. At room temperature, two signals are observed for the protons of the two...
-
On a particular day, there were 300 stocks that advanced on the NYSE and 800 that declined. The volume in advancing issues was 1000 and the volume in declining issues was 3000. What is the trin ratio?
-
A coupon bond that pays interest of $30 annually has a par value of $1000, matures in 6 years, and is selling today at $850. What is the yield to maturity on this bond?
-
A callable bond pays annual interest of $40, has a par value of $1000, matures in 4 years but is callable in 3 years at a price of $1100, and has a value today of $1020. What is the yield to call on...

Study smarter with the SolutionInn App


