Assign a name for each of the following compounds. Be sure to assign the configuration of each
Question:
a.
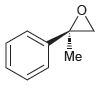
b.
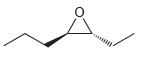
c.
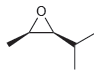
Transcribed Image Text:
Me
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
a S2phenyl1 2epoxypropane or S1me...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Assign a name for each of the following compounds. a. b. c.
-
Assign a name for each of the following compounds. a. b. c.
-
Assign a name for each of the following compounds: (a) (b) (c) (d) (e) (f) NH
-
Use the accompanying graph of y = f(x). Does exist? If it does, what is it? lim f(x)
-
Cheney Company sold a 20-ton mechanical draw press for $67,700. The old draw press cost $90,300 and had a book value of $62,000. Prepare the journal entry to record the disposition.
-
The world record for the \(100 \mathrm{~m}\) dash was set at 9.58 seconds a few years ago. What is this speed in kilometers per hour? In miles per hour?
-
A bar is \(4 \mathrm{~m}\) long and has a diameter of \(30 \mathrm{~mm}\). Determine the total amount of elastic energy that it can absorb from an impact loading if (a) it is made of steel for which...
-
Davis Corporation was authorized to issue 100,000 shares of $10 par common stock and 50,000 shares of $50 par, 6 percent, cumulative preferred stock. Davis Corporation completed the following...
-
Summaries models and methods of communications management in context of project life cycle and other project management functions. How would i include a real life example in to this answer?
-
Cisco Systems, Inc. (CSCO), manufactures and sells networking and communications equipment for transporting data, voice, and video and provides services related to that equipment. Its products...
-
a. Using the relationships derived in Example Problem 7.1 and the values of the critical constants for water from Table 7.2, calculate values for the van der Waals parameters a, b, and R from z c , T...
-
Identify the reactants you would use to form a racemic mixture of each of the following epoxides: a. b. c. d. Mery Me
-
Given an acceleration vector, initial velocity (u 0 , v 0 ), and initial position (x 0 , y 0 ), find the velocity and position vectors, for t 0. a(t) = (1,2), (uo, Vo) = (1, 1), (xo, yo) = (2,3)
-
How does a house account differ from other types of accounts? Give at least one example to explain your reasoning.
-
Brian contributes to his RRSP and RESP. Brian is the sole subscriber of the RESP for his son, Peter. Peter passed at the age of 15 due to an unexpected illness. At the time of death, the RESP was...
-
What is a performing art within humanities that makes you feel empathy?
-
Barry Allen owns interests in 3 partnerships: A, B, and C. He spends 600 hours a year on activities related to A, 75 hours on activities related to B and 50 hours on activities related to C. None of...
-
How is the right of women in Hinduism religion? What is Hinduism ?
-
Write an equation for the ellipse. e = 2/3 ; foci at (0, -9), (0, 9)
-
SBS Company have received a contract to supply its product to a Health Care Service Hospital. The sales involve supplying 1,250 units every quarter, the sales price is RM 85 per unit. The Client...
-
Order the following monomers with respect to their expected reactivity toward anionic polymerization, and explain your answer: H2C = CHCH3, H2C = CHC N, H2C = CHC6H5
-
Polystyrene is produced commercially by reaction ol styrene with butyl lithium as an anionic initiator. Using resonance structures, explain how the chain-carrying intermediate is stabilized.
-
Vinylidene chloride, H2C = CCl2, does not polymerize in isotactic, syndiotactic, and atactic forms. Explain.
-
The Devon Motor Company produces automobiles. On April 1, the company had no beginning inventories, and it purchased 6,290 batteries at a cost of $95 per battery. It withdrew 5,800 batteries from the...
-
Potter Industries has a bond issue outstanding with an annual coupon of 6% and a 10-year maturity. The par value of the bond is $1,000. If the going annual interest rate is 8.6%, what is the value of...
-
Varsity Furniture began June with merchandise inventory of 41 sofas that cost a total of $29,110. During the month, the company purchased and sold merchandise on account as follows: June 7 June 14...

Study smarter with the SolutionInn App


