Beta-keto esters can be prepared by treating the enolate of a ketone with diethyl carbonate. Draw a
Question:
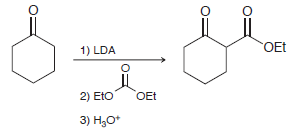
Transcribed Image Text:
OEt 1) LDA 2) Eto OEt 3) Н,о*
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (19 reviews)
O OEt OE...View the full answer

Answered By

SURAJ SINGH
I have completed my B.Tech in civil engineering.
I have done two year teacher training course.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A compound known as Hagemann's ester can be prepared by treating a mixture of formaldehyde and ethyl acetoacetate first with base and then with acid and heat. Write the structure for the product of...
-
-amino acids can be prepared by treating an aldehyde with ammonia and hydrogen cyanide, followed by acid-catalyzed hydrolysis. a. Give the structures of the two intermediates formed in this reaction....
-
Acrolein is an α,β-unsaturated aldehyde that is used in the production of a variety of polymers. Acrolein can be prepared by treating glycerol with an acid catalyst. Propose...
-
2. The long-term loan from World Bank had been taken out on 1 April 2018 to fund the company. According to the loan agreement, five annual installments of R36 000 are to be paid each year commencing...
-
A circular fin is made of pure copper with a thermal conductivity of k = 400 W / (m ( oC), h = 150 W / (m2 ( oC), mass density ( = 8900 kg / m3, and specific heat c = 375 J / (kg ( oC). The initial...
-
With perfect capital markets, as a firm increases its leverage, how does its debt cost of capital change? Its equity cost of capital? Its weighted average cost of capital?
-
Washington Mutual Insurance Company issued an \(\$ 80,000,7 \%, 10\)-vear bond payable at a price of 110 on January 1, 2009. Journalize the following transactions for Washington. Include an...
-
How do they relate to the difference between tax expense and taxes payable? How could an organization have a tax receivable? Why is the tax expense reported on the income statement comprised of...
-
Explain how the rows and columns created excel database would help you further analyze the information
-
Draw the structure of (2Z,5Z)-3-bromohepta-2,5-diene. Br A) B) Br C) D) Br Br
-
1. Describe Cream Silks promotion within the context of the multi attribute model: Which attribute(s) were central to the promotion and how does the model explain what they company was trying to...
-
Consider the three-variable linear regression model discussed in this chapter. a. Suppose you multiply all the X 2 values by 2. What will be the effect of this rescaling, if any, on the estimates of...
-
A particle P of mass 0.25 kg moves in a straight line on a smooth horizontal surface. P starts at the point O with speed 10ms -1 and moves towards a fixed point A on the line. At time is the...
-
What two indicators are used to classify performance measures?
-
1. What was the ASC? 2. What did it try to do? 3. When was it abandoned? 4. What took its place?
-
Susan Start, a new staff assistant of a CPA firm, was -assigned to the audit team auditing the financial statements of Rel-Hep Finance Company. The senior in charge of the audit assigned Susan to the...
-
How would you assess whether stock turnover and fixed asset turnover ratios were good or bad?
-
State the objectives in the audit of inventories.
-
Lopez acquired a building on June 1, 2012, for $1 million. Calculate Lopez's cost recovery deduction for 2018 if the building is: a. Classified as residential rental real estate. b. Classified as...
-
Consider the reaction of acetic acid in water CH 3 CO 2 H(aq) + H 2 O(l) CH3CO 22 (aq) + H 3 O + (aq) where Ka 5 1.8 3 1025. a. Which two bases are competing for the proton? b. Which is the stronger...
-
Explain the basicity order of the following three amines: p-nitroaniline (A), rz-nitroaniline (A), and aniline (C). The structures and pKa data are shown in Table 23'l'
-
Draw, at least two Fischer projections for the following molecules. (s) - 2butanol
-
Draw a conformational representation of: (a) -D-allopyranose (b) -idofiranose
-
Briefly describe the situation. Include a link to the story (optional). What issues specifically concern the organization, brand, product, or service? What communication/marketing channels did the...
-
3)The figure shows two wires that are tied to a 0.800 kg ball that revolves in a horizontal circle at a constant speed A) Draw the FBD and KD of the ball. If the tension in the top wire is 38.0 N, B)...
-
On January 1, 2025, Crane Inc. had these stockholders' equity balances. Common Stock, $1 par (2,200,000 shares authorized, 615,000 shares issued and outstanding) $615,000 Paid-in Capital in Excess of...

Study smarter with the SolutionInn App


